
Concept explainers
To analyze:
The inference that can be deciphered from the graph showing the relative fluidity of cell membranes in different species and whether the trend shown is apparent.
Given:
Researchers conducted an experiment to comparatively study the fluidity and composition of cell membranes. They maintained arctic sculpin at 0°C, a group of goldfish at 5°C, other group of goldfish at 25°C. They also maintained the desert pupfish at 34°C, and rats at normal temperature of 21°C. After keeping them for several days, the neuronal cells of all the animals were taken and membranes were isolated.
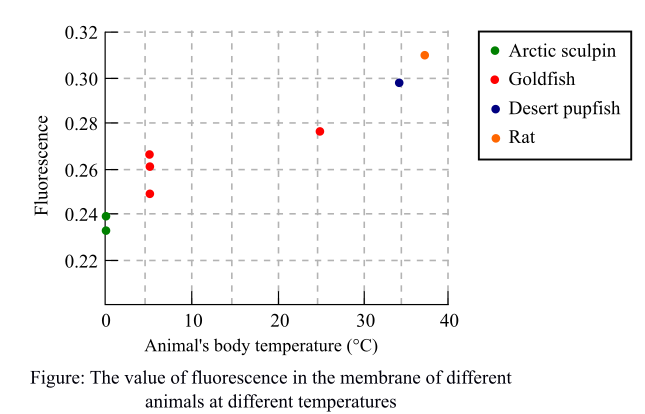
A fluorescent molecule was added to each of the extracted membranes and kept for some time at 20°C. Researchers then measured fluorescence and a graph was plotted by them. The graph depicted fluorescence against the body temperature of each of the animals.
The following graph shows the fluorescence of each animal depicted as colorful points at different temperatures. The more the value of fluorescence, the less will be the movement of molecules depicting a less fluid membrane.
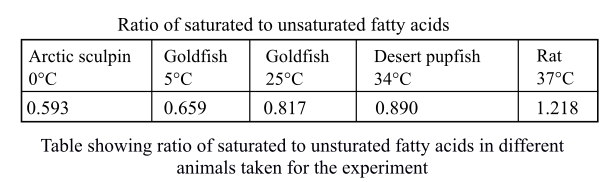
The following table was drawn by the researchers showing the ratio of saturated-to-unsaturated fatty acids in the phospholipid phosphatidyl choline for different animals taken into consideration.
Introduction:
The membrane is fluid in nature and is responsible for the transport of various molecules in and outside the cell. The fluidity of the membrane depends on the temperature and unsaturation. The low temperature leads to increase in fluidity to cope up the temperature. The saturated fatty acids are those fatty acids which do not have any double bond and have no scope of addition of any molecule. They can only substitute the molecules. The unsaturated fatty acids are those which have one or more double or triple bonds that increase the fluidity of the membrane. The species that survive in lower temperature have more amounts of unsaturated fatty acids so that they can avoid being frozen and remain fluid for transportation of the molecules.
Explanation of Solution
The data in the graph shows the value of fluorescence in different species at different temperature. It was observed that when the temperature of the animal’s body is low as in the case of arctic sculpin, the value of fluorescence is also low. There is an inverse relation between the movement of molecules and value of fluorescence. The low fluorescence depicts the high movement of molecules in the artic sculpin showing that the membrane has greater fluidity. The greater fluidity of the membrane helps these organisms to withstand such low temperature. In contrast, the fluidity of membranes of rats is low as the fluorescence value shown in the graph is high. This indicates that fluidity of membrane is not as high as in the species of low temperature.
The data of membrane fluidity depends on the temperature of the animal and is apparent as the fluidity of the membrane will change according to the change in the temperature.
Thus, it was concluded that the post translational modifications are important for the proper protein functioning and deoxyribonucleic acid (DNA) of the protein decides whether the modification should occur or not. The trend shown in the graph is apparent.
Want to see more full solutions like this?
Chapter 6 Solutions
EBK LIFE: THE SCIENCE OF BIOLOGY
- For the following scenarios, determine whether the molecules in the scenario are moving by simple diffusion (S), osmosis (0), facilitated diffusion (F), or active transport (A). a)For water to travel across the cell membrane at a substantial rate, the water molecules travel through protein channels known as aquaporins b)While water molecules are polar, they are also very small. Some water molecules are able to squeeze directly through the phospholipid bilayer due to their small size. C)Charged ions such as Na* travel through a cell membrane against their concentration gradient d)Cells lining the gut need to take in glucose, but at certain times, the concentration of extraceliular glucose is lower than the concentration already stored in the cells. e)At a certain time, glucose is in a high concentration outside of a cell and needs to travel through the membrane into the cell but does so using a glucose transporter protein.arrow_forwardSuppose the concentration of glucose inside a cell is 0.1 mM and the cell is suspended in a glucose solution of 0.01 mM. (a) What would be the free energy change involved in transporting 10-6 mole of glucose from the medium into the cell? Assume T = 37 °C. (b) What would be the free energy change involved in transporting 10-6 mole of glucose from the medium into the cell if the intracel- lular and extracellular concentrations were 1 mM and 10 mM, respectively? (c) If the processes described in parts (a) and (b) were coupled to ATP hydrolysis, how many moles of ATP would have to be hydrolyzed in order to make each process favorable? (Use the standard free energy change for ATP hydrolysis.)arrow_forwardA solution containing 3.58 x 1023 molecules/m3 of protein in water is separated from pure water by a membrane 3.20 μm thick. The diffusion coefficient of the protein through the membrane is 7.15 x 10-18 m2/s. On average, how many molecules cross 0.0240 μm2 of this membrane each second? [Answer as a positive number with 3 sig digits, but do not enter units with your answer]arrow_forward
- What is the value for the w (water potential) of a cell when placed in a hypertonic saline solution of 0.40 M under standard atmospheric pressure (1.0 MPa) and at a room temperature of 25° C? [Assume that the cell and solution , (solute potential) are at equilibrium. Use 0.08314L bar K mol as the gas constant (R)]arrow_forwardAn artificial cell consisting of an aqueous solution enclosed in a selectively permeable membrane (but with no cell wall) is immersed in a beaker containing an aqueous solution. The outside environment concentration consists of 0.01 M glucose and the inside of the cell has a concentration of 5.0 M glucose. The plasma membrane is permeable to water and monosaccharides, but impermeable to the disaccharides. Complete the following for the image below: Is the glucose going down or against its concentration gradient? Is the movement of the solute in the cell going out of the cell? Down...Yes Down...No Against...Yes Against...Noarrow_forwardExplain how salt concentrations contribute to the cell membrane intergrity in cells? What are the three different scenarios that could occur and why?arrow_forward
- In the experimental conditions described below, how many molecules of dextrose do you have to add to the extracellular fluid in order to make it iso-osmotic relative to the intracellular fluid? Intracellular fluid: Number of water molecules = 60 Number of Dextrose molecules = 5 Number of Sucrose molecules = 3 Extracellular fluid: Number of water molecules = 160 Number of dextrose molecule = ???? Number of Sucrose molecules = 0 Enter the number of molecules of dextrose in your answer (format: for 7 enter 7 or 7.00, for 7.5 enter 7.5 or 7.50, for 7.3333 enter 7.33 etc...).arrow_forwardA lab made cell consisting of an aqueous solution enclosed in a semipermeable membrane is immersed in a beaker containing a different aqueous solution. The cell membrane is permeable ONLY to water, glucose and fructose. What net movement across the cell membrane is expected if the solutions inside the cell and surrounding the cell have the concentrations described in the picture? Environment: "Cell"- 0.01 M sucrose 0.01 M glucose 0.01 M fructose 0.03 M sucrose 0.02 M glucose O fructose will move from the environment into the cell O glucose will move from inside the cell out O all of these are correct O fructose will move from inside the cell out O two these are correct O three of these are correct O sucrose will move from inside the cell out C F2 F3 F4 F5 F6 F7 F8 F9 F10 F11 F12 #3 & 关 4. 6 7 8 F 5 LL O o o oo O శ్రీarrow_forwardGiven the following cell type and solutions, draw the cell, showing the net direction of osmosis (water flow) and say what state the cell will be in. Choose from the following terms to describe the state of the cell: Turgid, flaccid, plasmolyzed, crenulated (shriveled), lysed (burst), normal. An animal cell with an internal osmolarity of 0.32 M (total concentration of all the solutes in solution) that is placed in a solution of 0.32 M NaCl.arrow_forward
- The following table depicts the rate of transport of a molecule into a cell as the external concentrations are changed. Based on what you know about the characteristics of membrane transport, the molecule is most likely transported by: a) passive transport b) simple diffusion across the membrane c) facilitated diffusion d) active transportarrow_forwardProvide 3 hypotheses about the fluidity and mosaicity of the cell membrane. Write the hypotheses in "IF-THEN" statement form. Example: If the detection tests result in a change of color, appearance, or pH value of the solution, then there is a movement of glucose, starch, NaCl, or H+ ions across the semi-permeable dialyzing membrane bag, and it is a good model.arrow_forwardSuppose the concentration of glucose inside a cell is 0.1 mM and the cell issuspended in a glucose solution of 0.01 mM.(a) What would be the free energy change, in kJ/mol, for the transport ofglucose from the medium into the cell? Assume T = 37 °C.(b) What would be the free energy change, in kJ/mol, for the transport ofglucose from the medium into the cell if the intracellular and extracellularconcentrations were 1 mM and 10 mM, respectively?(c) If the processes described in parts (a) and (b) were coupled toATP hydrolysis, how many moles of ATP would have to be hydrolysed,per mole of glucose transported, in order to make each process favorable? (Use the standard free energy change for ATP hydrolysis.)arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





