
ORGANIC CHEMISTRY LL PRINT UPGRADE
4th Edition
ISBN: 9781119810643
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 6, Problem 58IP
Interpretation Introduction
Interpretation:
The compound 1 is prepared and studied to investigate the intramolecular elimination mechanism. The curved arrows are to be interpreted for the given reaction mechanism:
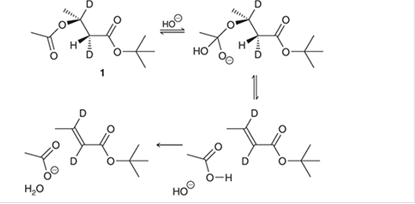
Concept introduction:
The chemical transformation can be shown by the arrow pushing method in which the curved arrows show the movement of electrons or the protons. In a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Write a detailed mechanism for the following reaction.
KCN
HCN
Trimethyl Phosphite reacts with bromomethane to afford the product shown below via a two-step mechanism. Draw a detailed mechanism for this reaction using arrows to show electron flow (both mechanistic steps are SN2 reactions).
Use the reaction given below to answer the following questions.
A
CH₂OH
B
H₂O+
C
+ CH3NH3*
D
a) What are the approximate pKas of compounds A and B?
b) The reaction given above is irreversible. Give a detailed explanation of the reaction and reaction
mechanism. Your explanation should indicate your knowledge of the reactivity of carboxylic acid
derivatives.
Chapter 6 Solutions
ORGANIC CHEMISTRY LL PRINT UPGRADE
Ch. 6.1 - Prob. 1LTSCh. 6.1 - Prob. 1PTSCh. 6.1 - Prob. 2ATSCh. 6.2 - Prob. 3CCCh. 6.3 - Prob. 4CCCh. 6.3 - Prob. 5CCCh. 6.4 - Prob. 6CCCh. 6.6 - Prob. 7CCCh. 6.7 - Prob. 2LTSCh. 6.7 - Prob. 8PTS
Ch. 6.7 - Prob. 9PTSCh. 6.7 - Prob. 10ATSCh. 6.8 - Prob. 3LTSCh. 6.8 - Prob. 11PTSCh. 6.8 - Prob. 12ATSCh. 6.9 - Prob. 4LTSCh. 6.9 - Prob. 13PTSCh. 6.9 - Prob. 14ATSCh. 6.10 - Prob. 5LTSCh. 6.10 - Prob. 15PTSCh. 6.10 - Prob. 16ATSCh. 6.11 - Prob. 6LTSCh. 6.11 - Prob. 17PTSCh. 6.11 - Prob. 18ATSCh. 6 - Prob. 19PPCh. 6 - Prob. 20PPCh. 6 - Prob. 21PPCh. 6 - Prob. 22PPCh. 6 - Prob. 24PPCh. 6 - Prob. 25PPCh. 6 - Prob. 26PPCh. 6 - Prob. 27PPCh. 6 - Prob. 28PPCh. 6 - Prob. 29PPCh. 6 - Prob. 30PPCh. 6 - Prob. 31PPCh. 6 - Prob. 32PPCh. 6 - Prob. 33PPCh. 6 - Prob. 34PPCh. 6 - Prob. 35PPCh. 6 - Prob. 36PPCh. 6 - Prob. 37PPCh. 6 - Prob. 38PPCh. 6 - Prob. 39PPCh. 6 - Prob. 40PPCh. 6 - Prob. 41PPCh. 6 - Prob. 43ASPCh. 6 - Prob. 44ASPCh. 6 - Prob. 45ASPCh. 6 - Prob. 46ASPCh. 6 - Prob. 47ASPCh. 6 - Prob. 48ASPCh. 6 - Prob. 49ASPCh. 6 - Prob. 50IPCh. 6 - Prob. 51IPCh. 6 - Prob. 52IPCh. 6 - Prob. 53IPCh. 6 - Prob. 54IPCh. 6 - Prob. 55IPCh. 6 - Prob. 56IPCh. 6 - Prob. 57IPCh. 6 - Prob. 58IPCh. 6 - Prob. 59IPCh. 6 - Prob. 60IPCh. 6 - Prob. 61IPCh. 6 - Prob. 62CPCh. 6 - Prob. 64CP
Knowledge Booster
Similar questions
- Propose a detailed mechanism for the transformations presented. represent the Intermediate structure A.arrow_forward5. The target compound below (a potential ẞ-blocker drug) may be prepared using a sulfonium ylide (and then a 2° amine) according to the retrosynthetic scheme shown below. Please perform the following: (a) Draw the structure of a suitable ylide that would accomplish this task and (b) write a mechanism to depict the formation and subsequent usage of the ylide (for the reaction in which the aldehyde is converted into the epoxide)...so the mechanism does NOT have to include the amine + epoxide part of the synthesis. HO NR2 1-8 potential class of ẞ-blocker drugs sulfonium ylide Harrow_forwardb) Synthesize the product on the right using any two organohalides of your choice (R-X, R'-X) with six carbon atoms or less in their structures. Describe the preparation of any organometallic species and cross-coupling conditions R-X R₁-Xarrow_forward
- Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction. The reaction involves the initial nucleophilic attack by diazomethane on the carbonyl carbon to form a tetrahedral intermediate. Collapse of this intermediate is accompanied by bond migration and loss of N,. For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. CH2N2, ether + N2 Draw the structure of the organic product(s) of the ring expansion of this compound: • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw one structure per sketcher Add additional ketchers usina the dron down menu in the bottom right co Previous Nextarrow_forwardProvide a reasonable mechanism for the transformation illustrated below.arrow_forwardDraw the major organic product for the reaction of this compound with PBr3. What type of mechanism is involved in this transformation?arrow_forward
- Predict the intermediate (A) and Reagent (B) in the reaction sequence below.arrow_forwardProvide the complete mechanism using Curved Arrow Formalism for the reaction shown below. State how many signals would be visualized in the 13C NMR spectrum for the product and give any pertinent IR data. 1. КОН, Н2О, heat, CH3(CH2),CN 2. HО → CH3(CH2),COHarrow_forwardProvide a detailed mechanism for each of the following reactions. a) b) c) سفی O=0 + NH,CH3 H₂O + CH3CH₂OH OH RiOH + ОН SOCI₂ H3O+ R CI + O=C H NHCH3 + H₂O SO₂ + HCIarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
