
ORGANIC CHEMISTRY LL PRINT UPGRADE
4th Edition
ISBN: 9781119810643
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6, Problem 37PP
Interpretation Introduction
Interpretation: The missing lone pairs and curved arrows is to be interpreted for the given mechanism.
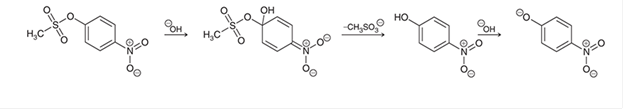
Concept introduction: In a
Electrophiles have a positive charge and are attracted toward an electronegative center. On the contrary, nucleophiles carry a negative charge and are attracted toward the electropositive center.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a curved arrow mechanism for the reaction. You can assume that all reactants and products are shown.
Add/Remove step
CN
:C=N:
Draw the structure of all products of the mechanism below.
H
:Br:
HHH
H
Click and drag to start
drawing a structure.
Draw curved arrows to show the movement of electrons in the step of the mechanism shown below.
Arrow-pushing Instructions
X→UU
:0:
:O:
||
CH3-C-CH₂-C-OEt
:OEt
:O:
||
:0:
||
CH3-C—CH—C—0—OEt + EtO:
H
Chapter 6 Solutions
ORGANIC CHEMISTRY LL PRINT UPGRADE
Ch. 6.1 - Prob. 1LTSCh. 6.1 - Prob. 1PTSCh. 6.1 - Prob. 2ATSCh. 6.2 - Prob. 3CCCh. 6.3 - Prob. 4CCCh. 6.3 - Prob. 5CCCh. 6.4 - Prob. 6CCCh. 6.6 - Prob. 7CCCh. 6.7 - Prob. 2LTSCh. 6.7 - Prob. 8PTS
Ch. 6.7 - Prob. 9PTSCh. 6.7 - Prob. 10ATSCh. 6.8 - Prob. 3LTSCh. 6.8 - Prob. 11PTSCh. 6.8 - Prob. 12ATSCh. 6.9 - Prob. 4LTSCh. 6.9 - Prob. 13PTSCh. 6.9 - Prob. 14ATSCh. 6.10 - Prob. 5LTSCh. 6.10 - Prob. 15PTSCh. 6.10 - Prob. 16ATSCh. 6.11 - Prob. 6LTSCh. 6.11 - Prob. 17PTSCh. 6.11 - Prob. 18ATSCh. 6 - Prob. 19PPCh. 6 - Prob. 20PPCh. 6 - Prob. 21PPCh. 6 - Prob. 22PPCh. 6 - Prob. 24PPCh. 6 - Prob. 25PPCh. 6 - Prob. 26PPCh. 6 - Prob. 27PPCh. 6 - Prob. 28PPCh. 6 - Prob. 29PPCh. 6 - Prob. 30PPCh. 6 - Prob. 31PPCh. 6 - Prob. 32PPCh. 6 - Prob. 33PPCh. 6 - Prob. 34PPCh. 6 - Prob. 35PPCh. 6 - Prob. 36PPCh. 6 - Prob. 37PPCh. 6 - Prob. 38PPCh. 6 - Prob. 39PPCh. 6 - Prob. 40PPCh. 6 - Prob. 41PPCh. 6 - Prob. 43ASPCh. 6 - Prob. 44ASPCh. 6 - Prob. 45ASPCh. 6 - Prob. 46ASPCh. 6 - Prob. 47ASPCh. 6 - Prob. 48ASPCh. 6 - Prob. 49ASPCh. 6 - Prob. 50IPCh. 6 - Prob. 51IPCh. 6 - Prob. 52IPCh. 6 - Prob. 53IPCh. 6 - Prob. 54IPCh. 6 - Prob. 55IPCh. 6 - Prob. 56IPCh. 6 - Prob. 57IPCh. 6 - Prob. 58IPCh. 6 - Prob. 59IPCh. 6 - Prob. 60IPCh. 6 - Prob. 61IPCh. 6 - Prob. 62CPCh. 6 - Prob. 64CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Add curved arrow(s) to draw the final step of the mechanism. H :ÖH H H CH, Edit Drawingarrow_forward2) Draw the complete curved arrow pushing mechanism for the following reactions. CO₂Et NaOH NH, CH3 I ✈ die 요 +arrow_forwardAdd curved arrow(s) to draw step 4 of the mechanism. Modify the given drawing of the product as needed to show the intermediate that is formed in this step. H 0 H + HÖ HC—Ở: :ö— Edit Drawing H₂CH. HO,arrow_forward
- Draw the simplest curved arrow mechanism possible for the reaction shown below. You may need to re-draw structures to show bond lines or lone pairs. D ~~ X AJ 'n harrow_forward4. Draw the mechanism for the following reaction using curved arrows. i NaBH4 CH3OH OHarrow_forwardDraw the full mechanism belowarrow_forward
- Draw the structure of all products of the mechanism below. H H H-C-C-C=C-H Br: Bri Click and drag to start drawing a structure.arrow_forwardDraw curved arrows for each step of the following mechanism: (SEE IMAGE)arrow_forwardPlease help with the following: Draw a detailed step-wise mechanism for the following reaction. Be sure to show all steps, intermediates, formal charges, and show the movement of electrons with curved arrows.arrow_forward
- Use curved arrow formalism to show the mechanism of the reaction shown belowarrow_forwardAdd curved arrow(s) to draw the final step of the mechanism. H₂O + HO Edit Drawing HO: Oarrow_forwardDraw a step-wise mechanism for the following substitution reaction. Be sure to add lone pairs and charges where relevant. CI H₂Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning