
Concept explainers
MATHEMATICAL For the following aspartase reaction (see Question 28) in the presence of the inhibitor hydroxymethylaspartate, determine
Interpretation:
The
Concept introduction:
In an enzymatic reaction, the inhibitors are the molecules that bind with the enzymes and block their activity. Enzymatic inhibition can be of many types. If a substrate and an inhibitor compete with each other to bind with the enzyme and both have similar structures, then the inhibition is known as competitive. On the other hand, if an inhibitor does not compete with the substrate and both of them can bind to an enzyme at their respective specific sites, then it is known as non-competitive inhibition.
Answer to Problem 61RE
Plot the reciprocal substrate concentration on the x-axis and two reaction velocities (both in the presence and absence of inhibitors) on the y-axis.
Before drawing the plot, the data needs to be modified so that it can be aligned in the Lineweaver–Burk plot. The modification of the data is as follows:
The Lineweaver–Burk plot from above data is:
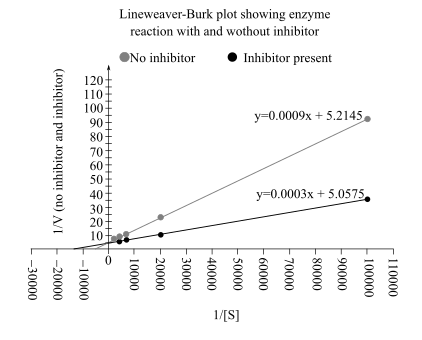
From this plot
In an uninhibited reaction, the
Therefore, the value of
Therefore, the value of
By comparing the Lineweaver–Burk plot of both reactions, it can be said that the inhibition is competitive in nature
Explanation of Solution
Given information:
In competitive inhibition, the substrate and the inhibitor have a similar structure. Hence, like the substrate, the inhibitor has also got nearly same affinity toward the active site of an enzyme and is able to bind with the enzyme. As a result, the enzyme’s affinity towards the substrate changes (it inhibits the substrate to bind with the enzyme). In other words, the
Also, in Lineweaver–Burk plot the
The reaction (without inhibitor) has a low value of
Want to see more full solutions like this?
Chapter 6 Solutions
Biochemistry
- REFLECT AND APPLY Noncompetitive inhibition is a limiting case in which the effect of binding inhibitor has no effect on the affinity for the substrate and vice versa. Suggest what a LineweaverBurk plot would look like for an inhibitor that had a reaction scheme similar to that on page 159 (noncompetitive inhibition reaction), but where binding inhibitor lowered the affinity of EI for the substrate.arrow_forwardREFLECT AND APPLY When we compare the binding of I and of S to the enzyme in a mixed noncompetitive inhibitor, we assumed that the binding of I decreased the affinity of the enzyme for S. What would happen if the opposite were true?arrow_forwardREFLECT AND APPLY What is the relationship between a transition-state analog and the induced-fit model of enzyme kinetics?arrow_forward
- REFLECT AND APPLY Why can we say that having a pure non- competitive inhibitor present is similar to just having less enzyme present?arrow_forwardREFLECT AND APPLY Would nature rely on the same enzyme to catalyze a reaction either way (forward or backward) if the DG were 0.8kcalmol1? If it were 5.3kcalmol1?arrow_forwardREFLECT AND APPLLY Is the reaction of 2-phosphoglycerate to phosphoenolpyruvate a redox reaction? Give the reason for your answer.arrow_forward
- MATHEMATICAL If a reaction can be written AB, and the G is 20kJmol1, what would the substrate/product ratio have to be for the reaction to be thermodynamically favorable?arrow_forwardREFLECT AND APPLY The enzyme D-amino acid oxidase has a very high turnover number because the D-amino acids are potentially toxic. The KM for the enzyme is in the range of 1 to 2 mM for the aromatic amino acids and in the range of 15 to 20 mM for such amino acids as serine, alanine, and the acidic amino acids. Which of these amino acids are the preferred substrates for the enzyme?arrow_forwardREFLECT AND APPLY Suggest a reason why a different reducing agent (NADPH) is used in anabolic reactions rather than NADH, which plays a role in catabolic ones.arrow_forward
- REFLECT AND APPLY Why is it somewhat misleading to study bio- chemical pathways separately?arrow_forwardREFLECT AND APPLLY What is the metabolic advantage of having both hexokinase and glucokinase to phosphorylate glucose?arrow_forwardMATHEMATICAL For an enzyme that displays MichaelisMenten kinetics, what is the reaction velocity, V (as a percentage of Vmax), observed at the following values? (a) [S]=KM (b) [S]=0.5KM (c) [S]=0.1KM (d) [S]=2KM (e) [S]=10KMarrow_forward
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
