
Concept explainers
(a)
Interpretation: The conformation which is present in higher concentration when
Concept introduction: The change in Gibbs free energy is represented by
If the
Answer to Problem 6.37P
The equatorial conformation is present in higher concentration in the given compound.
Explanation of Solution
Given:
The value of
The equilibrium reaction of monosubstituted cyclohexane is shown below.
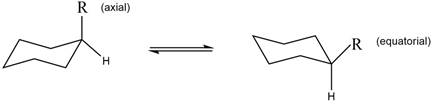
Figure 1
The value of
The equatorial conformation is present in higher concentration in the given compound.
(b)
Interpretation: The
Concept introduction: The change in Gibbs free energy is represented by
If the
Answer to Problem 6.37P
The
Explanation of Solution
The values of
The both given values are greater than
If the
Therefore, the
The
(c)
Interpretation: The
Concept introduction: The change in Gibbs free energy is represented by
If the
Answer to Problem 6.37P
The
Explanation of Solution
The values of
The equilibrium constant
Therefore, the
The
(d)
Interpretation: The
Concept introduction: The change in Gibbs free energy is represented by
If the
Answer to Problem 6.37P
The value of
Explanation of Solution
Given:
The values of
The relationship between
As the value of
The value of
(e)
Interpretation: The explanation corresponding to the relation of size of
Concept introduction: The change in Gibbs free energy is represented by
If the
Answer to Problem 6.37P
The large size of
Explanation of Solution
The value
The large size of
Want to see more full solutions like this?
Chapter 6 Solutions
ALEKS 360 CHEMISTRY ACCESS
- There are 17 possible alkene isomers with the formula C6H12. Draw structures of the five isomers in which the longest chain has six carbon atoms, and give the name of each. Are any of these isomers chiral? (There are also eight isomers in which the longest chain has five carbon atoms, and four isomers in which the longest chain has four carbon atoms. How many can you find?)arrow_forwardDraw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.arrow_forwardFor trans‑1‑ethyl‑4‑isopropylcyclohexane, which structures represent the possible boat conformations?arrow_forward
- Draw structural formulas for the cis and trans isomers of hydrindane. Show each ring in its most stable conformation. Which of these isomers is more stable?arrow_forwardDraw the structure of the following molecules, for which the name is given. 1. most stable conformation of 1,1-dichloro-2-bromoethane 2. N,N-Dimethylacetamide 3. cyclooctatetraene 4. bicyclo[2.2.1]heptane 5. spiro[2.4]heptanearrow_forwardConstruct the chair structures with the molecular models for trans-1-ethyl-3-methylcyclohexane. Find the energy of each conformer, the equilibrium constant Keq, and the percentage of each conformer at 25oC. Indicate which conformer is more stable.arrow_forward
- 3,4-Dimethylpent-1-ene has the formula CH2“CH¬CH(CH3)¬CH(CH3)2. When pure (R)-3,4-dimethylpent-1-ene is treated with hydrogen over a platinum catalyst, the product is (S)-2,3-dimethylpentane. The reactant is named (R), but the product is named (S). Does this name change imply a change in the spatial arrangement of the groups around the chiral center? So why does the name switch from (R) to (S)?arrow_forwardTrue or false Since the energy of the C-H bond is 416 kj/mol and the energy of the C=0rs bond is 678.5Kj/mol, it can be inferred that methanal is more reactive than methane.arrow_forwardThe sex attractant of the female tiger moth is an alkane of molecular formula C 18H 38. Is this molecule an acyclic alkane or a cycloalkane?arrow_forward
- Is isomerization of cis and trans -4-methyl-2-pentene possible at room temperature? Explain whyarrow_forwardWould there 5 isomers for hydrocarbons with the formula C4H8 ? ( including stereoisomersarrow_forwardDraw the staggered and eclipsed conformations that result from rotation around the C–C bond in CH3–CH2Br.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning




