
Rearrange the Clausius-Clapeyron equation, equation 6.14 in terms of the pressure
Interpretation:
The vapor pressures of
Concept introduction:
The Clausius-Clapeyron equation can be obtained from the rearrangement and integration of Clapeyron equation. The Clausius-Clapeyron equation is generally used for gas-phase equilibria, to predict the equilibrium temperatures and pressures and also for the determination of enthalpy for phase transition.
Answer to Problem 6.70E
The plot between vapor pressure and temperature for
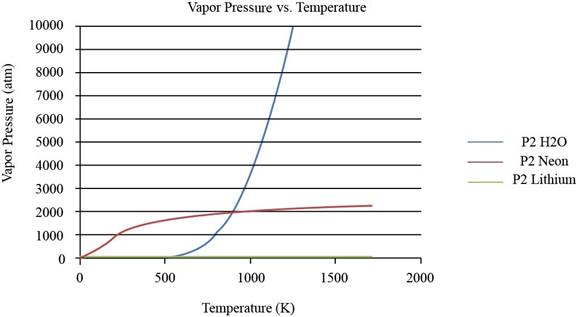
Figure 1
From the plot, the common observation is that for the given substances the increase in vapor pressure is slow until the normal boiling point.
Explanation of Solution
The Clausius-Clapeyron equation 6.14 is,
Rearrange the given equation for the partial pressure
Given boiling point of
Calculation of partial pressure
Calculation of partial pressure
Calculation of partial pressure
The plot between vapor pressure and temperature for
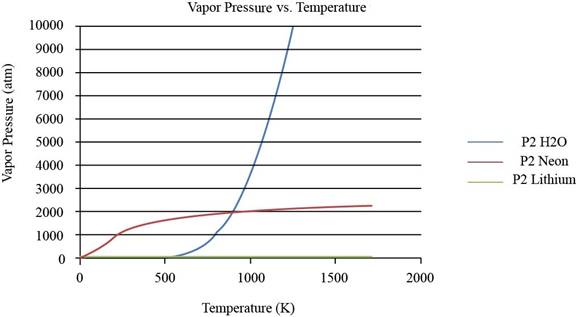
Figure 1
To observe the change in the plot of lithium, the values of vapor pressure is considered till
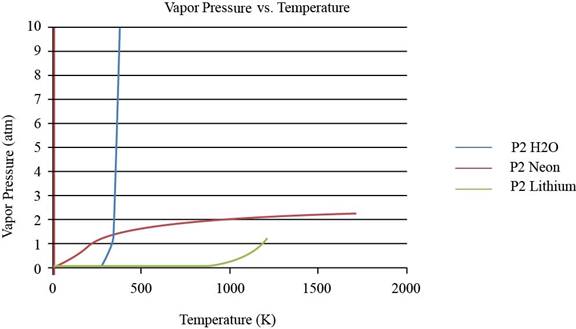
Figure 2
From the plot, the common observation is that for the given substances the increase in vapor pressure is slow until the normal boiling point. After the normal boiling point, the increase in vapor pressure is exponential. As the temperature increases from lower normal boiling point to higher values, the exponent value changes from negative to positive.
From the plot, the common observation is that for the given substances, the increase in vapor pressure is slow until the normal boiling point.
Want to see more full solutions like this?
Chapter 6 Solutions
Physical Chemistry
- Arrange the following substances in order of increasing strength of crystal forces: CO2, KCl, H2O, N2, CaO.arrow_forwardThe normal boiling point of SO2 is 263.1 K and that of NH3 is 239.7 K. At −40 °C, would you predict that ammonia has a vapor pressure greater than, less than, or equal to that of sulfur dioxide? Explain.arrow_forwardA 7.40 g sample of solid Ni(CN), ·4H,0 was heated such that the water turned to steam and was driven off. Assuming ideal behavior, what volume would that steam occupy at 1.00 bar and 100 °C? It may be useful to consult the periodic table. volume: Larrow_forward
- How much heat is required to convert 422 g of liquid H2O at 23.5 °C into steam at 150 °C?arrow_forwardThe melting point of copper metal is 1083 °C. The vapor pressure of solid copper is reported to be 3.33×10-7 atm at 1060 °C. Estimate the number of gaseous copper atoms per cubic centimeter in equilibrium with solid copper at 1060 °C (23 °C below its melting point). atoms/cm3arrow_forwardSulfur dioxide is produced in enormous amounts for sulfuric acid production. It melts at −73.0 ° C and boils at −10.0 ° C. Its Δ H o fus is 8.619 kJ/mol, and its Δ H o vap is 25.73 kJ/mol. The specific heat capacities of the liquid and gas are 0.995 J/g · K and 0.622 J/g · K, respectively. How much heat is required to convert 7.750 kg of solid SO2 at the melting point to a gas at 60.0 ° C?arrow_forward
- How much heat energy is required to convert 35.8 g of liquid sulfur dioxide, SO2, at 207.5 K to gaseous SO2 at 263.1K if the molar heat of vaporization of SO2 is 24.9 kJ/mol, and the specific heat capacity (?) of liquid SO2 is 1.36 J/(g·∘C)? Give q in kj.arrow_forwardAir conditioners dry the air while they cool the air. Suppose an air conditioner is used to cool a room that is 7.16 mx 10.60 m x 2.17 m when the outside temperature is 37 °C. At this temperature, the vapor pressure of water is 47.1 torr. The partial pressure of water in the air is 85.5% of the vapor pressure of water. How much water, in grams, is removed from the air each time the air from the room is cycled through the air conditioner? mass of H₂O: g A certain substance has a heat of vaporization of 33.26 kJ/mol. At what Kelvin temperature will the vapor pressure be 4.00 times higher than it was at 307 K? Karrow_forwardWhat amount of heat(in KJ) is required to convert 15.5G of an unknown liquiq(MM=83.21g/mol) at 12.9 degree Celsius to a gas at 90.4 degree Celsius? (Specific heat capacity of liquid =1.58J/g ; specific heat capacity of gas= .932 J/g; ΔHvap=22.5 kJ/mol; normal boiling point, Tb=57.3 degree Celsius)arrow_forward
- + 6. Give two examplea of two covalent solid which can undergo a solid-to-solid phase transition.arrow_forwardThe ambient temperature is 85.0°F and the humidity of the surrounding air is reported to be 67.0%. Using the Clausius-Clapeyron equation and the boiling point of water as 100.0°C at 760 torr, calculate the vapor pressure (in torr) of water in the air. Use 40.7 kJ/mol as the ∆Hvap of water.arrow_forwardWhat is the vapor pressure of SiCl4 in mmHg at 27.5∘C? The vapor pressure of SiCl4 is 100 mmHg at 5.4∘C, and ΔHvap = 30.2 kJ/mol.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning


