
(a)
Interpretation:
The
(a)
Explanation of Solution
The second deprotonation can be ignored.
The expression for equilibrium constant for the first deprotonation reaction can be written as shown below,
An equilibrium table can be set up as given below.
Now, these values in the fourth row can be inserted in the equilibrium constant expression as shown below.
Now, the above expression can be solved for x.
The
Therefore,
(b)
Interpretation:
The
(b)
Explanation of Solution
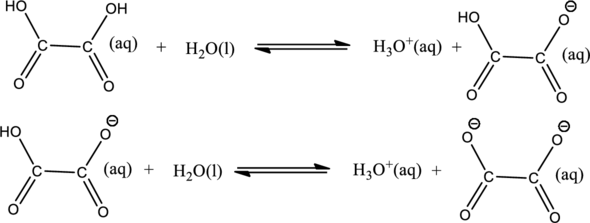
The value of
The second deprotonation can be ignored.
The expression for equilibrium constant for the first deprotonation reaction can be written as shown below,
An equilibrium table can be set up as given below.
Now, these values in the fourth row can be inserted in the equilibrium constant expression as shown below.
Now, the above expression can be solved for x.
The
Therefore,
(c)
Interpretation:
The
(c)
Explanation of Solution
The second deprotonation can be ignored.
The expression for equilibrium constant for the first deprotonation reaction can be written as shown below,
An equilibrium table can be set up as given below.
Now, these values in the fourth row can be inserted in the equilibrium constant expression as shown below.
Now, the above expression can be solved for x.
The
Therefore,
Want to see more full solutions like this?
Chapter 6 Solutions
CHEMICAL PRINCIPLES W/SAPLING
- Actually, the carbon in CO2(g) is thermodynamically unstable with respect to the carbon in calcium carbonate(limestone). Verify this by determining the standardGibbs free energy change for the reaction of lime,CaO(s), with CO2(g) to make CaCO3(s).arrow_forwardAdenosine triphosphate, ATP, is used to store energy within biological systems. It releases that energy by losing a terminal phosphate group, Pi, to form adenosine diphosphate, ADP, through the hydrolysis reaction ATP(aq) + H2O(I) → ADP(aq) + Pi(aq) + H+(aq) . The standard reaction Gibbs energy, ΔrGΘ, for the hydrolysis of ATP to ADP is+ 10 kJ mol-1 at 298 K.This assumes that the concentration of hydrogen ions is 1 mol dm-3 . (a) In biochemistry, it is common practice to assume an alternative , and more realistic, standard state for which pH = 7. What is the corresponding standard reaction Gibbs energy, denoted ΔrGΘ, for this biological standardstate? (b) What is the Gibbs energy of reaction in an environment at 37 °C and pH = 7 in which the ATP. ADP. and P; concentrations are all (i) 1.0 mmol dm-3, (ii) 1.0 μmol dm-3 ?arrow_forwardA constant current flows for 3.75 h through an electrolytic cell containing aqueous AgNO3(aq). During this time, 2.00 g of silver are deposited on the electrode. What is the current flowing through the cell?arrow_forward
- When acidulated water (dil.H2SO2solution) is electrolysed, will the pH of the solution be affected? Justify your answer.arrow_forwardOne ecologically important equilibrium is that between carbonate and hydrogencarbonate (bicarbonate) ions in natural water. (a) The standard Gibbs energies of formation of CO32−(aq) and HCO3−(aq) are −527.81 kJ mol−1 and −586.77 kJ mol−1, respectively. What is the standard potential of the HCO3−/CO32−,H2 couple? (b) Calculate the standard potential of a cell in which the cell reaction is Na2CO3(aq) + H2O(l) → NaHCO3(aq) + NaOH(aq). (c) Write the Nernst equation for the cell, and (d) predict and calculate the change in cell potential when the pH is changed to 7.0 at 298 K.arrow_forwardWhat volume of chlorine gas at standard temperature and pressure is evolved when a solution of MgCl2 is electrolyzed using a current of 12.4 A for 1.0 h?arrow_forward
- The standard potential of a Daniell cell, with cell reaction Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s), is 1.10 V at 25 °C. Calculate the corresponding standard reaction Gibbs energy.arrow_forwardThe standard cell potential of a Daniell cell, with cell reactionZn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s), is 1.10 V at 25 °C.Calculate the corresponding standard reaction Gibbs energy.arrow_forwardThe pH of the fluid in the human stomach following a meal is generally around 1.5. What is the hydrogen ion concentration in such a fluid?arrow_forward
- If the potential of a galvanic cell is +1.20 V, what is the Gibbs free-energy change when one mole of electrons is transferred in the oxidation–reduction reaction?arrow_forwardThe pH of an aqueous solution of 8.39×10-2 M ammonium nitrate, NH4NO3 (aq), isarrow_forwardBalance the following reactions. Calculate the cell potential and the Gibbs free energy of these reactions at standard conditions. Identify the reactions with K > 1 among the following redox reactions. Write the line representations for the Galvanic cells, indicating the anode and cathode.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning



