
(a)
Interpretation:
The products for the given reactions should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
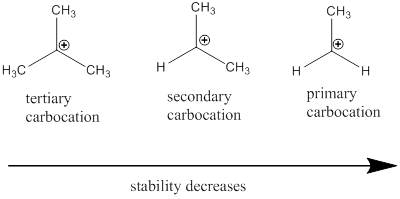
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
(b)
Interpretation:
The products for the given reaction should be determined.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of alkenes when two substituents are placed on same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Ozonolysis Reaction: It is an oxidative reaction which is used to oxidize the carbon-carbon double and triple bond.
(c)
Interpretation:
The reactivity of given set of compounds towards
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of alkenes when two substituents are placed on same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Ozonolysis Reaction: It is an oxidative reaction which is used to oxidize the carbon-carbon double and triple bond.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
ETEXT+MASTERINGCHEMISTRY STANDALONE AC
- Consider 3-iodo-2,3-dimethylpentane and 3-iodo-2methylpentane. a. which reacts faster in an Sn2 reaction? Explain. b. which reacts faster in an E2 reaction? Explain.arrow_forward1.Label kinetic product 2.Label the thermodynamic product 3.Predict the four products from reaction of alkene with one equivalent HBrarrow_forwarda. How many alkenes could you treat with H2, Pd/C to prepare methylcyclopentane? b. Which of the alkenes is the most stable? c. Which of the alkenes has the smallest heat of hydrogenation?arrow_forward
- What type of sigmatropic rearrangement is illustrated in each reaction?arrow_forwardRank the nucleophiles in each group in order of increasing nucleophilicity.a. -OH, -NH2, H2Ob. -OH, Br-, F- (polar aprotic solvent)c. H2O, -OH, CH3CO2-arrow_forwardRank the nucleophiles in each group in order of increasing nucleophilicity. a.−OH, −NH2, H2O b.−OH, Br−, F− (polar aprotic solvent) c.H2O, −OH, CH3CO2−arrow_forward
- What type of cycloaddition occurs in Reaction [1]? Draw the product of asimilar process in Reaction [2]. Would you predict that these reactionsoccur under thermal or photochemical conditions?arrow_forwardConsider the four trienes E–H.a.Rank compounds E–H in order of increasing heat of hydrogenation. b.Which compound is most reactive in the Diels–Alder reaction? c. Which compound(s) are unreactive in the Diels–Alder reaction? d.Which compound absorbs the longest wavelength of ultraviolet light?arrow_forward2-Methylpropane have more than one kind of hydrogen and will generate 2 different products with bromination in the presence of uv light. t or farrow_forward
- a. The reagent/s for REACTION 1 is/are ____ b. The reagent/s for REACTION 2 is/are ____ Choices: A. Sodium hydride and Chloromethane, B. Cyclopentanethiol and Br2, C. H2S, D. none of the choices c. The reaction mechanism for Reaction 1 is _____ Choices: A. SN1, B. SN2, C. SN Acyl, D. None of the choices d. The reaction mechanism for Reaction 2 is Choices: A. Oxidation, B. Reduction, C. Addition, D. None of the choicesarrow_forwardWhich of the following is product of A? How about B? a Cyclopentene oxide b 1,1-epoxycyclopentane c 1,2-hydroxycyclopentane d Cyclopentoxypentane What is the name of reaction 1? a hydration b hydrate formation c acid cleavage d epoxidation What is the name of reaction 2? a ring-opening reaction with Nu b hydration c Williamson ether synthesis d oxidationarrow_forwardConsider the reaction of (R)-2-chloro-3-methylbutane with sodium iodide to form aproduct.1(a) Draw the reaction scheme with the correct stereochemistry (reactant + NaI → product+ NaCl). Circle the nucleophile and draw a rectangle around the electrophile. 1(b) What is the symbol used for mechanism shown in 1(a)?1(c) If the sodium iodide was replaced with sodium hydroxide, the product is anALKENE. Draw a reaction MECHANISM to show how this happens.1(d) Draw the reaction energy diagram for the reaction in 1(c) and label the activationenergy.1(e) Using any alcohol with five carbons, and any carboxylic acid with six carbons, draw areaction to show how we would make an ester. 1(f) Describe the practical on esters.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





