
ETEXT+MASTERINGCHEMISTRY STANDALONE AC
7th Edition
ISBN: 9781269736947
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 6, Problem 90P
Interpretation Introduction
Interpretation:
The stereoisomer of alkene should be given which yields the (SS) and (RR) isomers of 3, 4-dimethyl-3-hexane, when it is reacts with
Concept Introduction:
Stereochemistry of addition reaction in
The syn addition of cis-alkene results the erythro (RS or SR) isomers and syn addition of trans-alkene results the threo (RR or SS) isomers.
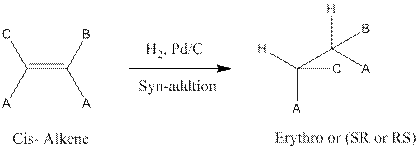
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which stereoisomer of 3-hexene forms (3S,4S)-4-bromo-3-hexanol and (3R,4R)-4-bromo-3-hexanol when it reacts with Br2 and H2O?
OChem HELP...
Draw the conformational structures (chair or boat conformations) for the MAJOR product formed when 1-tert-butylcyclohexene reacts with each of the following reagents. Also, indicate if the product obtained is racemic form.
a) Br2, CCl4
b) Br2, H2O
c) OsO4, then aqueous NaHSO3
d) ICl
e) mCPBA, then H3O+, H20
f) O3, then Me2S (conformational structure not required)
g) BH3:THF, then H2O2, HO-
h) D2, Pt
i) Hg(OAc)2 in THF-H2O, then NaBH4, HO-
j) BD3 :THF, then CH3CO2T
What organic product is formed when 1‑methylcyclopentene is treated with NMMO in the presence of H2O and a catalytic amound of OsO4? Clearly show stereochemistry by drawing a wedge and dashed bond for each chiral carbon.
Only draw one stereoisomer if more than one can be formed.
Chapter 6 Solutions
ETEXT+MASTERINGCHEMISTRY STANDALONE AC
Ch. 6.1 - Draw the mechanism for the reaction of cyclohexene...Ch. 6.2 - a. How many bond orbitals are avilable for...Ch. 6.2 - Prob. 3PCh. 6.2 - Prob. 4PCh. 6.3 - Prob. 5PCh. 6.4 - Prob. 6PCh. 6.4 - What alkene should be used to synthesize each of...Ch. 6.4 - Prob. 8PCh. 6.5 - The pKa of a protonated alcohol is about 2.5, and...Ch. 6.5 - Prob. 10P
Ch. 6.5 - Prob. 11PCh. 6.6 - a. What is the major product or each or the...Ch. 6.6 - Prob. 14PCh. 6.6 - Prob. 15PCh. 6.7 - What is the major product obtained from the...Ch. 6.8 - Which is more highly regionselective: reaction of...Ch. 6.8 - Prob. 19PCh. 6.9 - What will be the product of the preceding reaction...Ch. 6.9 - Prob. 21PCh. 6.9 - Prob. 22PCh. 6.9 - Prob. 23PCh. 6.9 - What is the product of the addition of 1Cl to...Ch. 6.9 - What will be the major product obtained from the...Ch. 6.9 - Propose a mechanism for the following reaction:Ch. 6.10 - Draw structures for the following: a. 24...Ch. 6.10 - What alkene would you treat with a peroxyacid in...Ch. 6.11 - What products are formed when the following...Ch. 6.11 - Prob. 30PCh. 6.11 - Prob. 31PCh. 6.12 - Prob. 34PCh. 6.12 - Prob. 35PCh. 6.13 - Prob. 36PCh. 6.13 - Prob. 37PCh. 6.13 - Prob. 38PCh. 6.14 - What characteristics must the reactant of a...Ch. 6.15 - Prob. 40PCh. 6.15 - What stereoisomers are obtained from each of the...Ch. 6.15 - Prob. 45PCh. 6.15 - Prob. 46PCh. 6.15 - Prob. 47PCh. 6.15 - Prob. 49PCh. 6.15 - Prob. 50PCh. 6.15 - Prob. 51PCh. 6.15 - Prob. 52PCh. 6.15 - Prob. 53PCh. 6.16 - Prob. 54PCh. 6.17 - Prob. 55PCh. 6.18 - Explain why 3-methykyclohexene should not be used...Ch. 6.18 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - What is the major product of the reaction of...Ch. 6 - Give two names for each of the following:Ch. 6 - Prob. 63PCh. 6 - Prob. 64PCh. 6 - Prob. 65PCh. 6 - What are the products of the following reactions?...Ch. 6 - When 3-methyl-1-butene reacts with HBr, two alkyl...Ch. 6 - Prob. 68PCh. 6 - Draw curved arrows to show the flow of electrons...Ch. 6 - What reagents are needed to carry out the...Ch. 6 - Prob. 71PCh. 6 - Prob. 72PCh. 6 - Prob. 73PCh. 6 - Prob. 74PCh. 6 - a. Draw the product or products that will be...Ch. 6 - The second-order rate constant (in units of M1s1)...Ch. 6 - Which compound has the greater dipole moment?Ch. 6 - Prob. 78PCh. 6 - Prob. 79PCh. 6 - Prob. 80PCh. 6 - Prob. 81PCh. 6 - Prob. 82PCh. 6 - Prob. 83PCh. 6 - Prob. 84PCh. 6 - Prob. 85PCh. 6 - Prob. 86PCh. 6 - Prob. 87PCh. 6 - Prob. 88PCh. 6 - Prob. 89PCh. 6 - Prob. 90PCh. 6 - 91. a. How many alkenes could you treat with H2,...Ch. 6 - Draw the products of the following reactions. If...Ch. 6 - Prob. 93PCh. 6 - Prob. 94PCh. 6 - Two chemists at Dupont found that lCH2Znl is...Ch. 6 - Prob. 96PCh. 6 - Prob. 97PCh. 6 - What alkene gives the product shown after...Ch. 6 - Prob. 99PCh. 6 - Prob. 100PCh. 6 - Prob. 101PCh. 6 - Prob. 102PCh. 6 - Prob. 103PCh. 6 - Propose a mechanism for the following reaction:Ch. 6 - Prob. 106PCh. 6 - Prob. 107P
Knowledge Booster
Similar questions
- Draw the product obtained when cis-2-butene is treated first with Br2 in CH2Cl2, second with NaNH2 in NH3, and then finally with Li in NH3.arrow_forwardWhich structure will not yield cis- or trans- isomers after a reaction with H2/Lindlar catalyst or Na/NH3?arrow_forwardDraw the product of the reaction of 1,2-dimethylcyclohexene with [math]H_{2}[/math] over a Pd catalyst. Show stereochemistry with wedged and dashed bonds.arrow_forward
- Two products are formed when methylenecyclohexane reacts with NBS? Show how each is formed. Disregard stereoisomers.arrow_forwardWrite the structure of the compound E,E-2,4-Hexadien-1-ol and label each non-equivalent carbon with a letter, A,B,C..arrow_forwardDraw all stereoisomers formed by monobromination of the cis andtrans isomers of 1,2-dimethylcyclohexane attached belowarrow_forward
- 8. Consider 1-bromo-2-methylpropane. Draw a Newman projection of the following (when viewed directlyalong the C1-C2 bond) a. staggered conformation of lowest energyb. eclipsed conformation of highest energyarrow_forwardHow many stereoisomers are formed from the reaction of 3-methylcyclohexene with NBS?arrow_forwardA chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactionsarrow_forward
- Account for the regioselectivity and stereoselectivity observed when 1-methylcyclopentene is treated with reagent. Q) BH3arrow_forwardDraw two diasteriomers of (1Z,4R)-1,4-dimethylcyclodecene and give the IUPAC name including E/Z and R/S notation.arrow_forwardDraw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecic.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning