
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
8th Edition
ISBN: 9780135213759
Author: John McMurry, David Ballantine
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6.1, Problem 6.4KCP
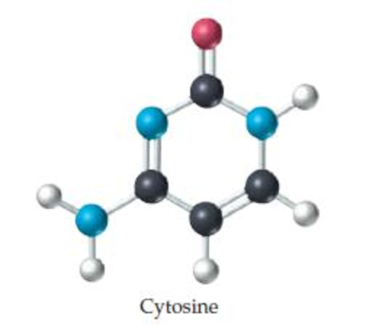
What is the molecular weight of cytosine, a component of DNA (deoxyribonucleic acid)? (black = C, blue = N, red = O, white = H.)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
If the GC content of a DNA molecule is 60%, what are the molar percentages of the four bases (G, C, T, A)?
Draw a structural formula for a nucleoside composed of the following.
Q.) b-2-Deoxy-d-ribose and cytosine
Draw a structural formula for a nucleoside composed of the following.
Q.) a-d-Ribose and adenine
Chapter 6 Solutions
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
Ch. 6.1 - Calculate the molecular weight of the following...Ch. 6.1 - How many molecules of ascorbic acid (vitamin C,...Ch. 6.1 - What is the mass in grams of 5.0 1020 molecules...Ch. 6.1 - What is the molecular weight of cytosine, a...Ch. 6.2 - Prob. 6.5PCh. 6.2 - Prob. 6.6PCh. 6.2 - Prob. 6.7PCh. 6.3 - Prob. 6.8PCh. 6.3 - Prob. 6.9PCh. 6.4 - Hydrogen fluoride is one of the few substances...
Ch. 6.4 - The tungsten metal used for filaments in light...Ch. 6.5 - What is the theoretical yield of ethyl chloride in...Ch. 6.5 - The reaction of ethylene oxide with water to give...Ch. 6.5 - Prob. 6.14KCPCh. 6.5 - Dietary iron forms a 1:1 complex with hemoglobin...Ch. 6.5 - Prob. 6.2CIAPCh. 6.5 - Prob. 6.3CIAPCh. 6 - Methionine, an amino acid used by organisms to...Ch. 6 - Prob. 6.16UKCCh. 6 - Prob. 6.17UKCCh. 6 - Prob. 6.18UKCCh. 6 - Prob. 6.19UKCCh. 6 - Prob. 6.20APCh. 6 - What is the difference between molecular weight...Ch. 6 - Prob. 6.22APCh. 6 - Prob. 6.23APCh. 6 - How many calcium atoms are in 16.2 g of calcium?Ch. 6 - What is the mass in grams of 2.68 1022 atoms of...Ch. 6 - Calculate the molar mass of each of the following...Ch. 6 - Prob. 6.27APCh. 6 - Prob. 6.28APCh. 6 - Caffeine has the formula C8H10N4O2. If an average...Ch. 6 - How many moles of aspirin, C9H8O4, are in a 500 mg...Ch. 6 - What is the molar mass of diazepam (Valium),...Ch. 6 - Calculate the molar masses of the following...Ch. 6 - How many moles are present in a 4.50 g sample of...Ch. 6 - How many grams are present in a 0.075 mol sample...Ch. 6 - The principal component of many kidney stones is...Ch. 6 - Prob. 6.36APCh. 6 - Ethyl acetate reacts with H2 in the presence of a...Ch. 6 - Prob. 6.38APCh. 6 - Ammonia, NH3, is prepared for use as a fertilizer...Ch. 6 - Hydrazine, N2H4, a substance used as rocket fuel,...Ch. 6 - Prob. 6.41APCh. 6 - Magnesium metal burns in oxygen to form magnesium...Ch. 6 - Titanium metal is obtained from the mineral...Ch. 6 - Prob. 6.44APCh. 6 - Prob. 6.45APCh. 6 - Prob. 6.47APCh. 6 - Prob. 6.48APCh. 6 - Once made by heating wood in the absence of air,...Ch. 6 - In Problem 6.40, hydrazine reacted with oxygen...Ch. 6 - Dichloromethane, CH2Cl2, the solvent used to...Ch. 6 - Cisplatin [Pt(NH3)2Cl2], a compound used in cancer...Ch. 6 - Prob. 6.53APCh. 6 - Prob. 6.54APCh. 6 - Prob. 6.55CPCh. 6 - Prob. 6.56CPCh. 6 - Prob. 6.57CPCh. 6 - Prob. 6.58CPCh. 6 - Prob. 6.59CPCh. 6 - Prob. 6.60CPCh. 6 - Gaseous ammonia reacts with oxygen in the presence...Ch. 6 - Sodium hypochlorite, the primary component in...Ch. 6 - Barium sulfate is an insoluble ionic compound...Ch. 6 - The last step in the production of nitric acid is...Ch. 6 - Acetylsalicylic acid, the active ingredient in...Ch. 6 - Jewelry and tableware can be silver-plated by...Ch. 6 - Elemental phosphorus exists as molecules of P4. It...Ch. 6 - Lithium oxide is used aboard the International...Ch. 6 - Prob. 6.69CPCh. 6 - Prob. 6.70GPCh. 6 - Obtain a bottle of aspirin and identify the amount...Ch. 6 - Lovastatin, a drug used to lower serum...Ch. 6 - Pyrite, also known as fools gold, is used...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- RNA is ribonucleic acid, and DNA is deoxyribonucleic acid. What exactly is deoxygenated about DNA?arrow_forwardWhat are the tautomeric forms of adenine and cytosine? of guanine, thymine, and uracil? What is the structure of the nucleotide called dCMP?arrow_forwardWhat tetrapeptide is synthesized from the informational DNA sequence G-T-C-A-G-T-A-C-G-T-T-A?arrow_forward
- Cytosine makes up 42% of the nucleotides in a sample of DNA from an organism. Approximately what percentage of the nucleotides in this sample will be thymine?arrow_forwardIf the G+C content of a sample of DNA is 46%, what are the proportions of all four nucleotides?arrow_forwardDNA molecules consist of chemically linked sequences of the bases adenine, guanine, cytosine, and thymine, denoted A, G, C, and T. A sequence of three basesiscalleda codon. A base may appear more than once in a codon. a) How many different codons are there? b) The bases A and G are purines, while C and T are pyrimidines. How many codons are there whose first and third bases are purines and whose second base is a pyrimidine? c) How many codons consist of three different bases?arrow_forward
- 2) When DNA is placed in distilled water, which is pH 7.0, it denatures (i.e., the two strands separate). The pH inside a cell is generally 7.2-7.5, depending on the organism, but DNA is generally double-stranded under physiological conditions. Briefly explain, in your own words, why DNA denatures when placed in distilled water but not when it is inside a cell. [Reminder: the pKa for the phosphate groups in the sugar-phosphate backbone of a strand of DNA is 2.14]arrow_forwardWhat are the monomers that makeup a nucleic acid and what are the monomers composed of? What type of bond is found along the backbone of the DNA backbone?arrow_forward5-Bromouridine is known to induce mutations in DNA. One of the characteristics of this compound is that the enol form is favored relative to the keto form. Draw the keto- and the eno- tautomers of the base. Determine (and draw) which base (A, T, G, or C) would most likely interact with each of the two forms by base-pair.arrow_forward
- (You can answer part (a) and part (c) together if it is more convenient to do so). DRAW condensed structural (or line-angle) formulas for the four nucleotides of DNA (A, C, G, T), and identify each with their actual names (not just A, C, G, or T).arrow_forwardAccording to Chargaff's rule of nitrogenous base pairing, which of the following statements is correct? If all adenine bonds to thymine and all cytosine pairs with guanine, then the sum of all adenine will never be equal to the sum of all thymine in a DNA molecule. If all adenine bonds to thymine and all cytosine pairs with guanine, then the sum of all adenine will never be equal to the sum of all thymine in an RNA molecule. If all adenine bonds to thymine and all cytosine pairs with guanine, then the sum of all adenine equals the sum of all thymine in a DNA molecule. If all adenine bonds to uracil and all cytosine pairs with guanine, then the sum of all adenine will never be equal to the sum of all uracil in an RNA molecule.arrow_forwardDNA structure depends on base pairing of its four nucleotides, A, C, T, and G. Nucleotide A pairs with T, and nucleotide C pairs with G. This forms a four-letter DNA “alphabet." Because DNA codes for amino acids in sets of three nucleotides, there are 4 cubed (4'), or 64, possible combinations, coding for 20 different amino acids. What is the best explanation for why there is no selective advantage for DNA to have five nucleotides (e.g., A, C, T, G, and E) with C pairing with either G or functionally equivalent E? It would be impossible to form the DNA molecule, because it must have an equal number of Cs and Gs. Because G and E have the same role, there would still be four functional letters of the alphabet. Replication would be inaccurate because sometimes C would bond with G and sometimes C would bond with E. There would be a five-letter alphabet with 125 combinations, which is too numerous. It is impossible because there are not five known nucleotides in the cell.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning

Human Heredity: Principles and Issues (MindTap Co...
Biology
ISBN:9781305251052
Author:Michael Cummings
Publisher:Cengage Learning
DNA Use In Forensic Science; Author: DeBacco University;https://www.youtube.com/watch?v=2YIG3lUP-74;License: Standard YouTube License, CC-BY
Analysing forensic evidence | The Laboratory; Author: Wellcome Collection;https://www.youtube.com/watch?v=68Y-OamcTJ8;License: Standard YouTube License, CC-BY