
Interpretation: A simplified sketch of the periodic table needs to be drawn and the blocks should be labeled s-, p-, d- and f-blocks.
Concept introduction: A periodic table consists of groups and periods. The horizontal rows are termed as groups and the vertical rows are termed as periods. The elements with similar properties are placed together in a group. Each period has different energy levels. Elements are also arranged according to various blocks in the periodic table such as s, p, d and f block.
Answer to Problem 15SSC
A simplified sketch of the periodic table is draw and s-, p-, d- and f-blocks are labeled.
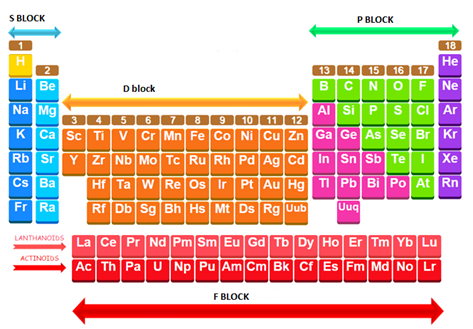
Group 1 and 2 are included in the s-block, Group 13-18 are included in the p-block, Group 3-12 are included in the d-block and F-block include the lanthanoids and the actinoids.
Explanation of Solution
Group 1 and 2 are included in the s-block. All the elements in this block has the general electronic configuration
Group 13-18 are included in the p-block. All the elements in this block has the general electronic configuration
Group 3-12 are included in the d-block. All the elements in this block has the general electronic configuration
F-block includes the lanthanide and actinide. Lanthanoids have the general electronic configuration
A simplified sketch of the periodic table with label the s-, p-, d- and f-blocks is given below:
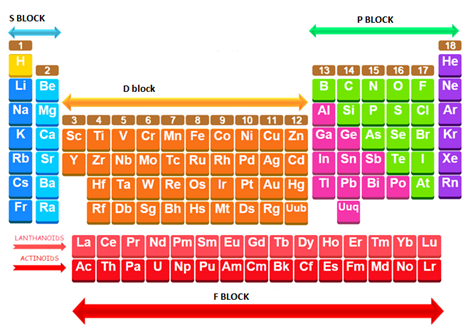
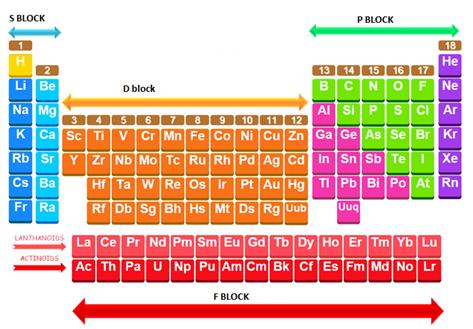
A simplified sketch of the periodic table and label the s-, p-, d- and f-blocks.
Chapter 6 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Inorganic Chemistry
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry
Chemistry: A Molecular Approach
Chemistry: The Central Science (13th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





