
EBK ORGANIC CHEMISTRY
6th Edition
ISBN: 9781260475685
Author: SMITH
Publisher: MCGRAW-HILL HIGHER EDUCATION
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 6.5, Problem 12P
The equilibrium constant for the conversion of the axial to the equatorial conformation of methoxycyclohexane is
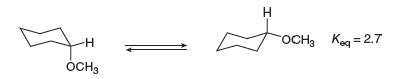
a. Given these data, which conformation is present in the larger amount at equilibrium?
b. Is
c. From the values in Table
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Difluoroacetylene (C2F2) gas has a ΔHf° = 241.2 kJ/mole and a a ΔGf° = 191.2 kJ/mole. Hexafluorobenzene(C6F6) gas has a ΔHf° = 132.8 kJ/mole and a a ΔGf° = 78.2 kJ/mole. For the following reaction:
C6F6(g) ⇌ 3C2F2(g)
Calculate:a) The ΔS for the reaction at 25 °Cb) The equilibrium constant K at 25 °Cc) The equilibrium constant K at 2700 °C (assuming there is no temperature dependance on ΔH and ΔS)
Consider these hydrocarbons: Butane, Hexane, Methane, Propane First look up the formulas, and write them down. Then put them in order from least entropy to most entropy. (Assume the temperature is hot enough that all are in gaseous form.)
For an addition reaction, why does free energy, ΔG, becomes more negative with decreasing temperature?
a. The negative enthalpy dominates at low temperature.
b. The negative entropy dominates at low temperature.
c. The positive entropy dominates at low temperature.
d. The positive enthalpy dominates at low temperature.
Chapter 6 Solutions
EBK ORGANIC CHEMISTRY
Ch. 6.2 - Prob. 2PCh. 6.3 - Problem 6.3 By taking into account...Ch. 6.3 - Problem 6.4 Use curved arrows to show the movement...Ch. 6.3 - Problem 6.5 Follow the curved arrows and draw the...Ch. 6.4 - Prob. 6PCh. 6.4 - Problem 6.7 Use the values in Table 6.2 to...Ch. 6.4 - Prob. 8PCh. 6.5 - aWhich Keq corresponds to a negative value of G,...Ch. 6.5 - Given each of the following values, is the...Ch. 6.5 - Given each of the following values, is the...
Ch. 6.5 - The equilibrium constant for the conversion of the...Ch. 6.6 - Prob. 13PCh. 6.6 - For a reaction with H=40kJ/mol, decide which of...Ch. 6.6 - For a reaction with H=20kJ/mol, decide which of...Ch. 6.7 - Draw an energy diagram for a reaction in which the...Ch. 6.7 - Prob. 17PCh. 6.7 - Prob. 18PCh. 6.8 - Problem 6.19 Consider the following energy...Ch. 6.8 - Draw an energy diagram for a two-step reaction,...Ch. 6.9 - Which value if any corresponds to a faster...Ch. 6.9 - Prob. 22PCh. 6.9 - Problem 6.23 For each rate equation, what effect...Ch. 6.9 - Prob. 24PCh. 6.10 - Identify the catalyst in each equation. a....Ch. 6 - Draw the products of homolysis or heterolysis of...Ch. 6 - Explain why the bond dissociation energy for bond...Ch. 6 - Classify each transformation as substitution,...Ch. 6 - Prob. 29PCh. 6 - 6.31 (a) Add curved arrows for each step to show...Ch. 6 - Prob. 35PCh. 6 - 6.39. a. Which value corresponds to a negative...Ch. 6 - Prob. 40PCh. 6 - For which of the following reaction is S a...Ch. 6 - Prob. 42PCh. 6 - Prob. 43PCh. 6 - 6.44 Consider the following reaction: .
Use curved...Ch. 6 - Prob. 45PCh. 6 - 6.50 The conversion of acetyl chloride to methyl...Ch. 6 - Prob. 50PCh. 6 - Prob. 53P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Given that the reaction of 4 NH3 (g) + 5 O2 (g) → 4 NO (g) + 3 H20 (g) ΔΗ - - 906 kJ What would AH for NO (g) + ? H20 (g) → NH3 (g) + O2 (g) be? 2. 906 kJ 226.5 kJ -226.5kJ 453 kJarrow_forwardP4(s) + 5O2(g) → P4O10(s) ∆ G° = -2697.0 kJ/mol2H2(g) + O2(g) → 2h2O(g) ∆ G° = - 457.18 kJ/mol 6H2O(g) + P4O10(s) → 4H3PO4(1) ∆G° = - 428.66 kJ/mol What is the standard free energy of formation, ∆ Gf degree, for phosphoric acid? a: 11243.3 kj/mol b: -11243.3 kj/mol c: 4497.2 kj/mol d: -4497.2 kj/molarrow_forwardQuestion:In a chemical reaction, a compound X undergoes a transformation to form compound Y. The reaction is carried out at constant temperature and pressure. Compound Y is observed to have a higher energy content than compound X. Explain this phenomenon based on the principles of thermodynamics.arrow_forward
- Part I. Free Energy - What's Up With That? 1. Write out the equation for Free Energy and define what each letter represents. 2. Where is energy stored in organic molecules? What term in the equation above represents this stored potential energy in a molecule? 3. Every chemical reaction loses some energy to entropy, every time. Briefly explain how making entropy smaller or larger effects the free energy in a reaction?arrow_forwardFQ 29: Predict the enthalpy, entropy, and free energy change for reactions. ta a) Describe the enthalpy change for this reaction step. b) Describe the entropy change for this reaction step. c) Describe the free energy change for this reaction step at 25 °C. hanisms for thearrow_forward1. Which of the following is correct? * The change in free energy is the total free energy of the products added to the total free energy of the reactants. The change in free energy is the total free energy of the reactants added to the total free energy of the products. The change in free energy is the total free energy of the products subtracted from the total free energy of the reactants. The change in free energy is the total free energy of the reactants subtracted from the total free energy of the products. 2. A reaction is an equilibrium if... The change in the free energy of the system is less than zero. The change in the free energy of the system is zero. Change in free energy of the system is greater than zero. The reaction quotient is less than the equilibrium constant.arrow_forward
- 1. The condensation of glutamate and ammonia to yield glutamine and water has a AG'° of +14.23 kJ/mol. Depict this reaction in its thermodynamically favorable direction.arrow_forwardCells use the hydrolysis of adenosine triphosphate (ATP) as a source of energy. The conversion of ATP to ADP has a standard free-energy change of -30.5 kJ>mol. If all the free energy from the metabolism of glucose, C6H12O61s2 + 6 O21g2 ¡ 6 CO21g2 + 6 H2O1l2 goes into the conversion of ADP to ATP, how many moles of ATP can be produced for each mole of glucose?arrow_forwardThe molar heat of fusion (∆fusH) for acetone is 5.7 kJ/mol and the molar heat of vaporization (∆vapH) for acetone is 31.3 kJ/mol. At 1.0 bar pressure, acetone melts at -95 oC and boils at 56 oC. Calculate the entropy change for boiling 1 mole of acetone at 56oC.arrow_forward
- What is the Born-Haber process and how is it important to the world’s economyarrow_forwardA. Suggest chemical tests to differentiate the following pairs of organic compounds given the chemical reaction for the positive sign. Write the observable change for the positive sign of reaction and the chemical reaction for the compound that will test positive. Chemical Test and Positive Sign of Reaction Test: Compound Chemical Reaction which will Test for the Positive Positive Sign он H3C H3C° CH3 Positive Sign: Sw1 halogenation `CH3 2-pentanone Он 2-pentanol Test: H3C haloform formation H3C `CH3 Positive Sign: isopropyl alcohol 1-propanolarrow_forward6H2O + 6CO2 --> C6H12O6 + 6O2 H=+2803 kJ/mol calculate the amount of sunlight energy required (in kJ) by a plant to synthesize 75.0g of glucose (C6H12O6)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY