
Concept explainers
a)
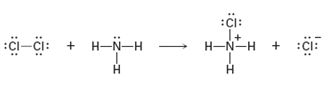
Interpretation:
Curved arrows are to be added to the polar reaction given to indicate the flow of electrons.
Concept introduction:
Curved arrows are drawn such that they start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained. The formation of a new bond will be accompanied by the breaking of an existing bond.
To add:
Curved arrows to the polar reaction given for indicating the flow of electrons.
b)
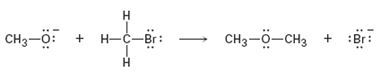
Interpretation:
Curved arrows are to be added to the polar reaction given to indicate the flow of electrons.
Concept introduction:
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To add:
Curved arrows, to the polar reaction given, to indicate the flow of electrons.
c)
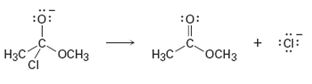
Interpretation:
Curved arrows are to be added to the polar reaction given to indicate the flow of electrons.
Concept introduction:
Curved arrows start from a nucleophilic source (neutral or negatively charged) and end in an electrophilic sink (neutral or positively charged). During the flow of the electrons the octet rule must be maintained both in the source and sink.
To add:
Curved arrows, to the polar reaction given, to indicate the flow of electrons.
Trending nowThis is a popular solution!

Chapter 6 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- An electrostatic potential map of calicene is shown below. a) Both the electrostatic potential map and its significant dipole moment indicate that calicene is an unusually polar hydrocarbon. Which of the dipolar resonance forms, structure A or structure B, better corresponds to the electron distribution in the molecule? Select the single best answer. b) Which one of the following structures should be stabilized by resonance to a greater extent? Select the single best answer.arrow_forwardComplete the equation for the reaction between the following Lewis acid-base pair. Use curved arrows to show the flow of electrons in the reaction and draw the product. Assign lone pairs and radical electrons where appropriate. Apply formal charges where appropriate. • Draw the appropriate electron-flow arrows. • Use the "starting points" menu to revert to the original molecule(s) shown. • Omit + signs between structures. ● / CH3 1- H₂C-C CH3 H در St ? ChemDoodleⓇarrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided resonance structures, draw the curved electron-pushing arrows to show the interconversion between resonance hybrid contributors. Be sure to account for all bond-breaking and bond-making steps. Drawing Arrows N ▸ Qarrow_forward
- Draw thee other resonance structures for the following carbocation. Use electronpushing arrows in order to derive one resonance structure from another. Indicate theformal charge on each atom in your structures.arrow_forwardWhich sets of curved arrows accounts for the protonation of propene with HI?arrow_forwardComplete the equation for the following proton-transfer reaction by using curved arrows to show the flow of electron pairs and drawing the products of the reaction. • Draw all atoms, including hydrogen atoms. Apply formal charges where appropriate. Assign lone pairs and radical electrons where appropriate. • Use the "starting points" menu to revert to the original molecule(s) shown. • Draw the appropriate electron-flow arrows. • Omit + signs between structures. ● ● H-O: == starting points == ↑ TAYY H T H-C-H H-N-H T H در ? ChemDoodlearrow_forward
- Select the single best answer. Considering only electron density, will the following reaction occur? CI CH30: + yes noarrow_forwardConsider the acid-base reaction below to answer the following question(s). + H-Q-H - CH₂ base acid A) Using the curved arrow formalism, show the flow of electrons for this reaction. B)Write the products of this reaction. CH,CH-arrow_forwardIn the following acid - base reactions, a) Draw Lewis structures of the reactants and the products. b) Determine which species are acting as electrophiles (acids) and which are acting as nucleophiles (bases). c) Use the curved - arrow formalism to show the movement of electron pairs in these reactions and the imaginary movement in the resonance hybrids of the products. d) Indicate which reactions are best termed Brønsted-Lowry acid - base reactions i. CH3CHO + HCI-- > CH3CH2O + + Cl- ii. CH3CHO + OH- - - > CH3CO-(OH) Harrow_forward
- Complete the equation for the following proton-transfer reaction by using curved arrows to show the flow of electron pairs and drawing the products of the reaction. • Draw all atoms, including hydrogen atoms. • Apply formal charges where appropriate. Assign lone pairs and radical electrons where appropriate. • Use the "starting points" menu to revert to the original molecule(s) shown. • Draw the appropriate electron-flow arrows. • Omit + signs between structures. 0▾ HH HH == starting points == V H-CI: کر ChemDoodle ?arrow_forwardwhich of the following structures is different from the other three?arrow_forward6. (Chapter 15-Q37) Indole is an aromatic heterocyclic that has a benzene ring fused to a pyrrole ring. Answer the following questions. Indole 6(a) What is the hybridization of N in this molecule? = 6(b) How many pi electrons N contributes to the ring? = 6() Which orbitals contribute to form a sigma bond between N and H in this molecule? = 6(c) What is the electronic relationship of Indole to naphthalene? Give the answer by comparing number of rings and number of pi electrons in both compounds, write x rings, y pi electrons=|arrow_forward
