
ORGANIC CHEMISTRY-EBOOK>I<
9th Edition
ISBN: 9781305084414
Author: McMurry
Publisher: INTER CENG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.SE, Problem 47AP
The naturally occurring molecule α-terpineol is biosynthesized by a route that includes the following step:
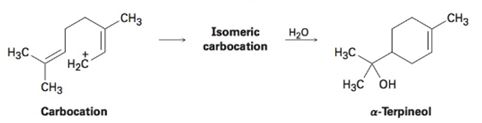
(a) Propose a likely structure for the isomeric carbocation intermediate.
(b) Show the mechanism of each step in the biosynthetic pathway, using curved arrows to indicate electron flow.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Suppose we aempt the conversion of fumaric acid to deuterated malic acid with BD3·THF,followed by oxidation with D2O2in NaOD(aq). Show all the possible stereoisomers (as Fisherprojections) that may be formed, and draw the mechanistic pathways (showing stereochemistry)that lead to these possible products
Following is a balanced equation for bromination of toluene.(a) Using the values for bond dissociation enthalpies given in Appendix 3,calculate ∆H0for this reaction.(b) Propose a pair of chain propagation steps and show that they add up to theobserved reaction.(c) Calculate ∆H0for each chain propagation step.(d) Which propagation step is rate-determinin
Q1
Chapter 6 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
Ch. 6.1 - Prob. 1PCh. 6.3 - Prob. 2PCh. 6.3 - Using curved fishhook arrows, propose a mechanism...Ch. 6.4 - Prob. 4PCh. 6.4 - An electrostatic potential map of boron...Ch. 6.5 - What product would you expect from reaction of...Ch. 6.5 - Reaction of HBr with 2-methylpropene yields...Ch. 6.6 - Prob. 8PCh. 6.6 - Predict the products of the following polar...Ch. 6.7 - Which reaction is more energetically favored, one...
Ch. 6.7 - Prob. 11PCh. 6.9 - Which reaction is faster, one with ∆G‡ = +45...Ch. 6.10 - Prob. 13PCh. 6.SE - Prob. 14VCCh. 6.SE - Prob. 15VCCh. 6.SE - Prob. 16VCCh. 6.SE - Look at the following energy diagram: (a) Is...Ch. 6.SE - Look at the following energy diagram for an...Ch. 6.SE - What is the difference between a transition state...Ch. 6.SE - Prob. 20EDRMCh. 6.SE - Prob. 21EDRMCh. 6.SE - Draw an energy diagram for a two-step exergonic...Ch. 6.SE - Draw an energy diagram for a reaction with keq =...Ch. 6.SE - The addition of water to ethylene to yield ethanol...Ch. 6.SE - When isopropylidenecyclohexane is treated with...Ch. 6.SE - Prob. 26EDRMCh. 6.SE - Draw the electron-pushing mechanism for each...Ch. 6.SE - Draw the complete mechanism for each polar...Ch. 6.SE - Prob. 29EDRMCh. 6.SE - Identify the functional groups in the following...Ch. 6.SE - Identify the following reactions as additions,...Ch. 6.SE - Identify the likely electrophilic and nucleophilic...Ch. 6.SE - For each reaction below identify the electrophile...Ch. 6.SE - Prob. 34APCh. 6.SE - Follow the flow of electrons indicated by the...Ch. 6.SE - Prob. 36APCh. 6.SE - Prob. 37APCh. 6.SE - Despite the limitations of radical chlorination of...Ch. 6.SE - Prob. 39APCh. 6.SE - Answer question 6-39 taking all stereoisomers into...Ch. 6.SE - Prob. 41APCh. 6.SE - Prob. 42APCh. 6.SE - Prob. 43APCh. 6.SE - The reaction of hydroxide ion with chloromethane...Ch. 6.SE - Prob. 45APCh. 6.SE - Ammonia reacts with acetyl chloride (CH3COCl) to...Ch. 6.SE - The naturally occurring molecule α-terpineol is...Ch. 6.SE - Prob. 48APCh. 6.SE - Prob. 49APCh. 6.SE - Draw the structures of the two carbocation...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the products of spherification of the following acid / alcohol pairsarrow_forwardDraw structures corresponding to the following names: (a) 3-hydroxyhexanoic acid (b) 2-iodo-2-methyloctanoic acid (c) 2-butynoic acid (d) 5-ethyl-6-oxoheptanoic acid (e) o-Hydroxybenzoic acid (f) cis-3-isopropylcyclohexanecarboxylic acidarrow_forwardA key step in the hydrolysis of acetamide in aqueous acid proceeds by nucleophilic addition of * OH (a) H3O* to CH3Ĉ NH2 (b) H2O to CH3ČNH2 + OH +OH (c) H3O* to CH,ČNH2 (d) HO¯ to CH3CNH2arrow_forward
- (a) How many Stereoisomers are possible for 9,10- dibromoexadecanoic acid?(b) Addition of bromine to palmitoleic acid primarily yields a set of enantiomers, (±) -three-9,10-dibromohexadecanoic acid. The addition of bromine is an anti-addition to the double bond (ie, it appears to occur through the intermediary of the bromonium ion). Taking into account the stereochemistry of the cis double bond of palmitoleic acid and the stereochemistry of adding bromine,Write a three-dimensional structure for (±) -three-9,10-dibromohexadecanoic acid!arrow_forward(b) 3-methyl-2-butanol reacts with concentrated sulphuric acid to form 2-methyl-2- butene. Write the mechanism for the reaction.arrow_forward6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward
- Draw and name the seven aldehydes and ketones with the formula C5H10O. Which are chiral?arrow_forward(c)Show step by step how to synthesize methoxybenzene from benzene.arrow_forward(a) A hydrocarbon isolated from fish oil and from plankton was identified as 2,6,10,14-tetramethyl-2-pentadecene. Write its structure.(b) Alkyl isothiocyanates are compounds of the type RN C S. Write a structural formula for allyl isothiocyanate, a pungent-smelling compound isolated from mustard.(c) Grandisol is one component of the sex attractant of the boll weevil. Write a structural formula for grandisol given that R in the structure shown is an isopropenyl group.arrow_forward
- (а) Н (Б) Н (c) H Each of the monomers shown here can undergo free radical polymerization. For each polymerization: (i) Show the mechanism for the first two propagation steps, using benzoyl peroxide as the initiator. (ii) Draw the condensed formula for the polymer, showing the repeating unit. (iii) Provide a name for the polymer. H H H CI Hi c=0 H OCH3 H,N Vinylidene chloride Acrylamide Methyl vinyl etherarrow_forwardPlease draw the skeletal formula of the following compounds: (A) Isobutyraldehyde (B) α-Ethyl-γ-methoxycaproaldehyde (C) 6-Hydroxyhexanal (D) 2,4-Pentanedione (E)3-Cyano-7-oxoheptanoic acidarrow_forwardAspirin, or 2-acetoxybenzoic acid, (C9H8O4) is often synthesised from salicylic acid.(i) Sketch and discuss any changes in the number of possible structural conformations ofaspirin relative to those of salicylic acid. (ii) Re-draw the structure predicted to be the lowest energy conformation of aspirin,indicating any expected stabilising and destabilising interactions. Justify your choice.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY