
Refer to Example 7-5 dealing with the work done by 0.100 mol He at 298 K in expanding in a single step from 2.40 to 1.20 atm. Review also the two-step expansion
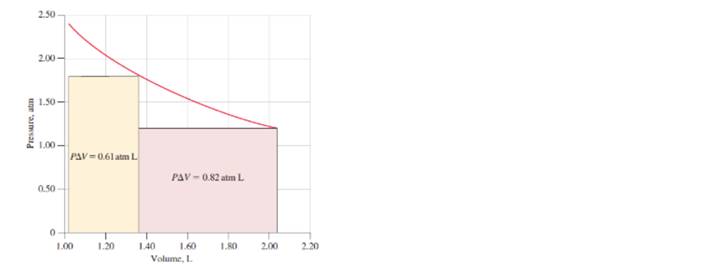
a. Determine the total work that would be done if the He expanded in a series of steps, at 0.10atm intervals, from 2.40 to 1.20 atm.
b. Represent this total work on the graph below, in which the quantity of work done in the two-step expansion is represented by the sum of the colored rectangles.
c. Show that the maximum amount of work would occur if the expansion occurred in an infinite number of steps. To do this, express each infinitesimal quantity of work as
d. Imagine reversing the process, that is, compressing the He from 1.20 to 2.40 atm. What are the maximum and minimum amounts of work required to produce this compression? Explain.
e. In the isothermal compression described in part (d), what is the change in internal energy assuming ideal gas behavior? What is the value of q?
f. Using the formula for the work derived in part (c), obtain an expression for
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
GENERAL CHEMISTRY(LL)-W/MASTERINGCHEM.
- 9.11 Analyze the units of the quantity (pressurevolume) and show that they are energy units, consistent with the idea of PV-work.arrow_forward9.12 PV-work occurs when volume changes and pressure remains constant. If volume is held constant, can PV-work be done? What happens to Equation 9.2 when volume is held constant? E=q+w (9.2)arrow_forwardOne statement of the second law of thermodynamics is that heat cannot be turned completely into work. Another is that the entropy of the universe always increases. How are these two statements related?arrow_forward
- Would the amount of heat absorbed by the dissolution in Example 5.6 appear greater, lesser, or remain the same if the heat capacity of the calorimeter were taken into account? Explain your answer.arrow_forward9.54 The phase change between graphite and diamond is difficult to observe directly. Both substances can be hurned, however. From these equations, calculate H for the conversion of diamond into graphite. C(s,graphite)+O2(g)CO2(g) H= -393.51 kJ C(s,diamond)+O2(g)CO2(g) H= -395.94 kJarrow_forwardA 220-L cylinder contains an ideal gas at a pressure of 150 atm. If the gas is allowed to expand against a constant opposing pressure of 1.0 atm, how much work is done? The expansion will stop when the internal pressure equals the external pressure. Use Boyles law to determine the final volume.arrow_forward
- Explain in your own words what is meant by the term entropy. Explain how both matter spread and energy spread are related to the concept of entropy.arrow_forwardCoal is used as a fuel in some electric-generating plants. Coal is a complex material, but for simplicity we may consider it to be a form of carbon. The energy that can be derived from a fuel is sometimes compared with the enthalpy of the combustion reaction: C(s)+O2(g)CO2(g) Calculate the standard enthalpy change for this reaction at 25C. Actually, only a fraction of the heat from this reaction is available to produce electric energy. In electric generating plants, this reaction is used to generate heat for a steam engine, which turns the generator. Basically the steam engine is a type of heat engine in which steam enters the engine at high temperature (Th), work is done, and the steam then exits at a lower temperature (Tl). The maximum fraction, f, of heat available to produce useful energy depends on the difference between these temperatures (expressed in kelvins), f = (Th Tl)/Th. What is the maximum heat energy available for useful work from the combustion of 1.00 mol of C(s) to CO2(g)? (Assume the value of H calculated at 25C for the heat obtained in the generator.) It is possible to consider more efficient ways to obtain useful energy from a fuel. For example, methane can be burned in a fuel cell to generate electricity directly. The maximum useful energy obtained in these cases is the maximum work, which equals the free-energy change. Calculate the standard free-energy change for the combustion of 1.00 mol of C(s) to CO2(g). Compare this value with the maximum obtained with the heat engine described here.arrow_forwardNitrogen gas reacts with hydrogen gas to form ammonia gas .Consider the reaction between nitrogen and hydrogen as depicted below: a. Draw what the container will look like after the reaction has gone to completion. Assume a constant pressure of 1 atm. b. Is the sign of work positive or negative, or is the value of work equal to zero for the reaction? Explain your answer.arrow_forward
- A piston initially contains 688 mL of gas at 1.22 atm. What work is done if the piston expands against a constant external pressure of 733 torr? The expansion will stop when the internal pressure equals the external pressure. Use Boyles law to determine the final volume.arrow_forwardCalculate the work donewhen 1.000 mole of an ideal gas expands reversiblyfrom 1.0L to 10L at 298.0K. Then, calculatethe amount ofwork done when the gasexpands irreversibly against aconstant external pressure of 1.00 atm. Compare thetwovaluesand comment.arrow_forwardFor the reaction NO(g)+NO2(g)N2O3(g) , use tabulated thermodynamic data to calculate H and S. Then use those values to answer the following questions. (a) Is this reaction spontaneous at 25°C? Explain your answer. (b) If the reaction is not spontaneous at 25°C, will it become spontaneous at higher temperatures or lower temperatures? (c) To show that your prediction is accurate, choose a temperature that corresponds to your prediction in part (b) and calculate G . (Assume that both enthalpy and entropy are independent of temperature.)arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





