
Concept explainers
(a)
Interpretation: The name of the given structure needs to be determined.
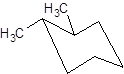
Concept Introduction: Chair conformation is the most stable conformation of cyclohexane. It is represented as follows:

Here, the substituted groups in the chair conformation are represented as follows:
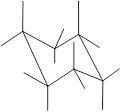
The groups showing in straight upward and downward directions are axial and the groups bend slightly right or left are equatorial.
The axial and equatorial groups are represented in the chair conformation as A and E:
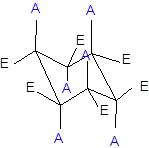
(b)
Interpretation: The given structure on the right needs to be completed which is obtained by chair flip.
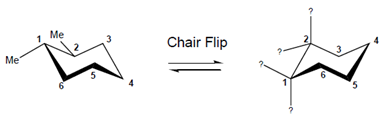
Concept Introduction:After a chair flip, all the axial bonds become equatorial and all the equatorial bonds become axial. But, the groups in the up direction remains upward and groups in down direction remain downward.
Also, if the molecule is cis, it remains cis even after chair flip. The same is the case with trans molecule.
(c)
Interpretation: The name of the structure formed after the chair flip in part (b) needs to be determined.
Concept Introduction: Chair conformation is the most stable conformation of cyclohexane. It is represented as follows:

Here, the substituted groups in the chair conformation are represented as follows:
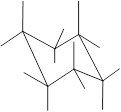
The groups showing in straight upward and downward directions are axial and the groups bend slightly right or left are equatorial.
The axial and equatorial groups are represented in the chair conformation as A and E:
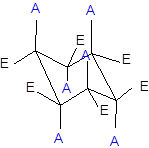
(d)
Interpretation: Whether the two structures are consistent with the given facts about chair flip or not needs to be determined.
Concept Introduction:
The chair conformation of cyclohexane is represented as follows:

During the flipping, no bond is break. The numbering in the chair form is represented as follows:
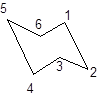
During ring flipping, mirror image of the chair conformation is formed.
It is represented as follows:
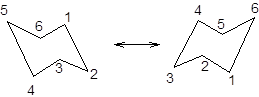
Trending nowThis is a popular solution!

Chapter 7 Solutions
Custom eBook for Organic Chemistry
- A student suggests that the molecule on the right can be made from a single molecule with one less ring in it. If the student is correct, draw the starting material below, otherwise, check the box under the drawing area. ? + NaOH Click and drag to start drawing a structure. olo Ararrow_forwardA. Draw the major resonance structure, then draw curved arrows to show how this can be converted to the Lewis structure given. B. Thionitromethane is redrawn for you. Draw curved arrows to show how this can be converted to the resonance structure you drew in part A.arrow_forwardShown below is Streptomycin, and Neomycin B. Circle and label as many functional groups in these molecules as you can. a. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? b. Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?arrow_forward
- Must answer completearrow_forwardALL ONE QUESTION THANKS Make a line and 3d model of each molecule below A.) What type of bonds are in each molecule B.) which bonds allow rotationarrow_forwardConstruct a model in which a tetrahedral carbon atom has four different colored model atoms attached to it- red, green, orange and white representing 4 different atoms attached to the central atom. a) Does the atom have a plane of symmetry? why or why not? b) Now replace the green atom in your model with a second orange atom. Now two of the groups attached to the carbon atom are identical. Does the model now have a plane of symmetry? Describe it. c)A carbon atom has four different groups attached to the stereogenic center. Draw structural formulas for the following compound and mark stereogenic centers with as asterisk: 1-bromobutane, 2-bromobutane, 1,2-dibromobutane, 1,4-dibromobutane, 2,3-dibromobutane.arrow_forward
- Complete the mechanism of the given enamine to imine tautomerization by adding any missing atoms, bonds, charges, nonbonding electron pairs, and curved arrows. Then, indicate which of the species will predominate at equilibrium. NH-R is a generic acid with conjugate base :NH,-R in solution. Note the resonance arrows in the scheme. Step 1: add curved arrows. Step 2: complete the structure and add curved arrows. Select Draw Rings Groups More Erase Select Draw Rings Groups More Erase C H N /|の +) H,N -R H,N - R 1L H H. H H Harrow_forwardPlease helparrow_forwardSection A. Check your understanding + 'H' P-H 1. Explain why a carbonyl C=O bond is protonated at the lone pair while and alkene C-C bond is protonated at the pi bond. Use a MO diagram to justify your answer. C-C-Harrow_forward
- Build a model of 2,2,5,5-tetramethylhexane. Orient the model so that you are looking at the carbon with the arrow pointing to it in Figure 3. Align the bond to the next carbon in the chain so that it is directly behind the first carbon to match a Newman projection view. (See Figure MM.3 in the lab manual) Spin the carbons on either side of the bond you're looking down to cycle through all three staggered and all three eclipsed positions of the substituents. Draw all six positions as Newman projections on the data sheet and identify the position with the highest energy. Draw the six Newman projections of all of the different energy levels. Label each as staggered or eclipsed and rank in order from lowest energy to highest.arrow_forward1. Nucleophile and Electrophile Identification a. Which of the following species is likely to behave as a in the box below each species. electrophile? Put your answer nucleophile and which as an CH3O NH4 NO2 Species CN Br Nuc or Elect? CH H-C C-H НО" Species CH3S NH3 Nuc or Elect? b. Draw the following Functional Groups, including lone pairs of electrons. Predict the bond polarity of the functional group using S+ and &- if applicable (think about electronegativity differences!). Then, identify ALL nucleophilic and electrophilic sites using your knowledge of bond polarity. EXAMPLE: Alcohol Aldehyde Amine Acid Chloride Hас-о-н electrophilic electrophilic nucleophilic Alkene Ketone Ether Carboxylic Acidarrow_forwardc. Complete each acid/base reaction, determine the favored direction of the equilibrium, and explain (short answer). CI CI CI OH ONa NaOH d. Show the two Newman projections (looking down Cl-C2, C5-C4) and indicate any gauche interactions) for following molecule. Br "CI e. Circle and name the functional groups in the following molecule. OH f. Show the orbital energy level diagram of the hybridized oxygen atom in the following structure; indicate hybridization, and the number of o and z bonding orbitals.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
