
Concept explainers
To estimate: The K value from the given data that describes the ligand binding to a protein.
Concept introduction: Dissociation constant is the equilibrium constant at which the ligand dissociates from the protein. It is denoted as K. Y is fractional saturation of the ligand occupying the ligand-binding site in the protein, or it can be defined as the number of ligands that has dissociated from the protein. K is achieved when fractional saturation is half at a particular ligand concentration.
Answer to Problem 1E
Answer: The dissociation constant is estimated by plotting the values in a graph and finding the ligand concentration when Y is 0.5 (half of fractional saturation).
Pictorial representation:
Fig.1 shows a graph with ligand (mM) in the X-axis and Y in the Y-axis.
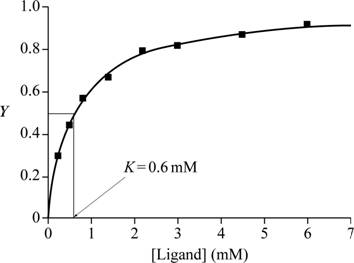
Fig.1: Graph of ligand (mM) vs Y to estimate K.
Explanation of Solution
Explanation:
When fractional saturation is half, it means that the ligand has occupied half of the ligand binding site in the protein. When fractional saturation is half, half of the ligand is bound to half the number of proteins in the medium. Half of proteins and half of ligands are free in the medium. This can be called as the equilibrium state of the ligand-protein binding reaction, and thus K is obtained at half of the fractional saturation.
A graph is plotted with ligand concentration in mM on the X-axis and Y, the fractional saturation, on the Y-axis with the given data. At half of fractional saturation, that is, at equilibrium state, the corresponding ligand concentration is 0.6 Mm, which is evident from the graph.
Conclusion: From the graph plotted for the given values, the K value is 0.6mM.
Want to see more full solutions like this?
Chapter 7 Solutions
FUNDAMENTALS OF BIO(LL)W/ACCESS>CUSTOM<
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





