
Concept explainers
(a)
Interpretation: The systematic name and common name of the given organic compound is to be interpreted.
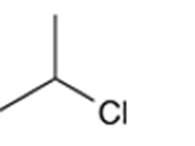
Concept Introduction: Organic compounds are chemical compounds that are mainly composed of carbon and hydrogen atoms. The naming of organic compounds is followed the IUPAC rules. The longest chain of carbon atoms is considered the parent chain. The substituents must be given with the least locant number. The locant number must be written as the prefix in the IUPAC name.
(a)
Answer to Problem 47PP
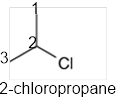
The systematic name of the given compound must be 2-chloropropane and the common name is isopropyl chloride.
Explanation of Solution
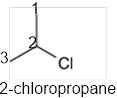
In the given organic compound, the parent longest chain contains 3 carbon atoms so the root word must be propane with one substituent; chloro at C2 position. Thus, the systematic name of the given compound must be 2-chloropropane and the common name is isopropyl chloride.
(b)
Interpretation: The systematic name and common name of the given organic compound is to be interpreted.
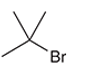
Concept Introduction: Organic compounds are chemical compounds that are mainly composed of carbon and hydrogen atoms. The naming of organic compounds is followed the IUPAC rules. The longest chain of carbon atoms is considered the parent chain. The substituents must be given with the least locant number. The locant number must be written as the prefix in the IUPAC name.
(b)
Answer to Problem 47PP
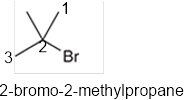
The systematic name of the given compound must be 2-bromo-2-methylpropane and the common name is tert-butyl bromide.
Explanation of Solution
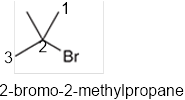
In the given organic compound, the parent longest chain contains 3 carbon atoms so the root word must be propane with two substituents: chloro and methyl at 2 positions. Thus, the systematic name of the given compound must be 2-bromo-2-methylpropane and the common name is tert-butyl bromide.
(c)
Interpretation: The systematic name and common name of the given organic compound is to be interpreted.

Concept Introduction: Organic compounds are chemical compounds that are mainly composed of carbon and hydrogen atoms. The naming of organic compounds is followed the IUPAC rules. The longest chain of carbon atoms is considered the parent chain. The substituents must be given with the least locant number. The locant number must be written as the prefix in the IUPAC name.
(c)
Answer to Problem 47PP
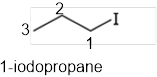
The systematic name of the given compound must be 1-iodopropane and the common name is propyl iodide.
Explanation of Solution
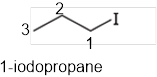
In the given organic compound the parent longest chain contains 3 carbon atoms so the root word must be propane with one substituent; iodo at 1 position. Thus, the systematic name of the given compound must be 1-iodopropane and the common name is propyl iodide.
(d)
Interpretation: The systematic name and common name of the given organic compound is to be interpreted.
Concept Introduction: Organic compounds are chemical compounds that are mainly composed of carbon and hydrogen atoms. The naming of organic compounds is followed the IUPAC rules. The longest chain of carbon atoms is considered the parent chain. The substituents must be given with the least locant number. The locant number must be written as the prefix in the IUPAC name.
(d)
Answer to Problem 47PP
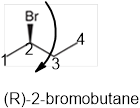
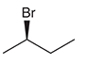
The systematic name of the given compound must be (R)-2-bromobutane and the common name is (R)- sec-butyl bromide.
Explanation of Solution
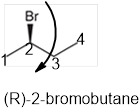
In the given organic compound the parent longest chain contains 4 carbon atoms so the root word must be butane with one substituent; bromo at C2 position. Thus, the systematic name of the given compound must be (R)-2-bromobutane and the common name is (R)- sec-butyl bromide. The prefix (R) indicates the clockwise rotation of the groups bonded at the chiral C atom when arranged from 1 to 4 in the increasing order of their
(e)
Interpretation: The systematic name and common name of the given organic compound is to be interpreted.
Concept Introduction: Organic compounds are chemical compounds that are mainly composed of carbon and hydrogen atoms. The naming of organic compounds is followed the IUPAC rules. The longest chain of carbon atoms is considered the parent chain. The substituents must be given with the least locant number. The locant number must be written as the prefix in the IUPAC name.
(e)
Answer to Problem 47PP
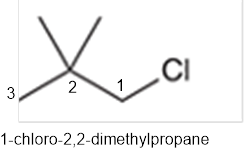
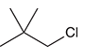
The systematic name of the given compound must be 1-chloro-2,2- dimethylpropane and common name is neopentyl chloride.
Explanation of Solution
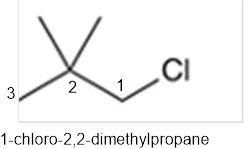
In the given organic compound, the parent longest chain contains 3 carbon atoms so the root word must be propane with three substituents as methyl and chloro at C1 and C2 position. Thus, the systematic name of the given compound must be 1-chloro-2,2- dimethylpropane and the common name is neopentyl chloride.
(f)
Interpretation: The systematic name and common name of the given organic compound is to be interpreted.
Concept Introduction: Organic compounds are chemical compounds that are mainly composed of carbon and hydrogen atoms. The naming of organic compounds is followed the IUPAC rules. The longest chain of carbon atoms is considered the parent chain. The substituents must be given with the least locant number. The locant number must be written as the prefix in the IUPAC name.
(f)
Answer to Problem 47PP
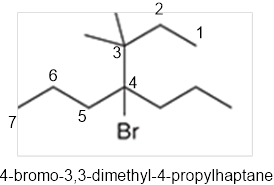
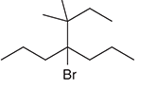
The systematic name of the given compound must be 4-bromo-3,3-dimethyl-4-propylhaptane.
Explanation of Solution
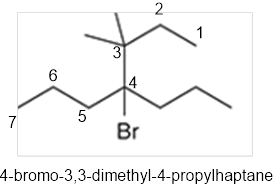
In the given organic compound the parent longest chain contains 7 carbon atoms so the root word must be haptane with four substituents as propyl, methyl and bromo. Thus, the systematic name of the given compound must be 4-bromo-3,3-dimethyl-4-propylhaptane.
Want to see more full solutions like this?
Chapter 7 Solutions
ORGANIC CHEMISTRY
- Arrange the following compounds in increasing order of their property as indicated :(i) CH3COCH3, C6H5COCH3, CH3CHO(reactivity towards nucleophilic addition reaction)(ii) Cl—CH2—COOH, F—CH2—COOH, CH3—COOH (acidic character)arrow_forwardDraw as many compounds as you can that fit the following descriptions:arrow_forwardDraw the structural formulas for the following compounds. Include all the bonds to hydrogen atoms. Be sure to answer both parts. (a) 1,2,4-trimethylbenzene: (b) chlorobenzene:arrow_forward
- (b) Draw the structural formula for each of the following compounds. Lukiskan formula struktur bagi setiap sebatian berikut. (i) 3-fluoro-2,4,4-trimethylpent-2-ene (ii) ethyl butanoate (ii) 2,4-dichloro-3-methylpentanoic acidarrow_forwardOrganic Chemistry: Nomenclature for Alkyl Halides Give a systematic name for the following compound:arrow_forwardGive the systematic name of the compound below.arrow_forward
- An unknown organic compound is found on elementalanalysis to contain 68.1% carbon, 13.7% hydrogen, and18.2% oxygen by mass. It is slightly soluble in water. Uponcareful oxidation it is converted into a compound thatbehaves chemically like a ketone and contains 69.7% carbon,11.7% hydrogen, and 18.6% oxygen by mass. Indicatetwo or more reasonable structures for the unknown.arrow_forwardWhat's the name of this compound ? Assign a systematic name for the following compound:arrow_forwardLl.171. Subject:- Chemistryarrow_forward
- Calculate the enthalpy of formation of propane from:a. Combustion data (ΔHc = –2217 kJ mol–1; ΔHf (CO2) = –393.4 kJ mol–1, ΔHf (H2O) =–285.2 kJ mol–1);b. Bond enthalpies (C–C 348 kJ mol–1; C–H 413 kJ mol–1; H–H 436 kJ mol–1; ΔHvap forcarbon = –717 kJ mol–1).arrow_forwardPlease be clear in your writing The following names may have some errors. Correct the name and render the structures corresponding to the following names. g) 1,3-pentadiino h) cyclohexylacetylenearrow_forwardProvide the best possible systematic name for the following compound:arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning



