
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 6TYK
Use the following U-tube setup to answer questions 6 through 8.
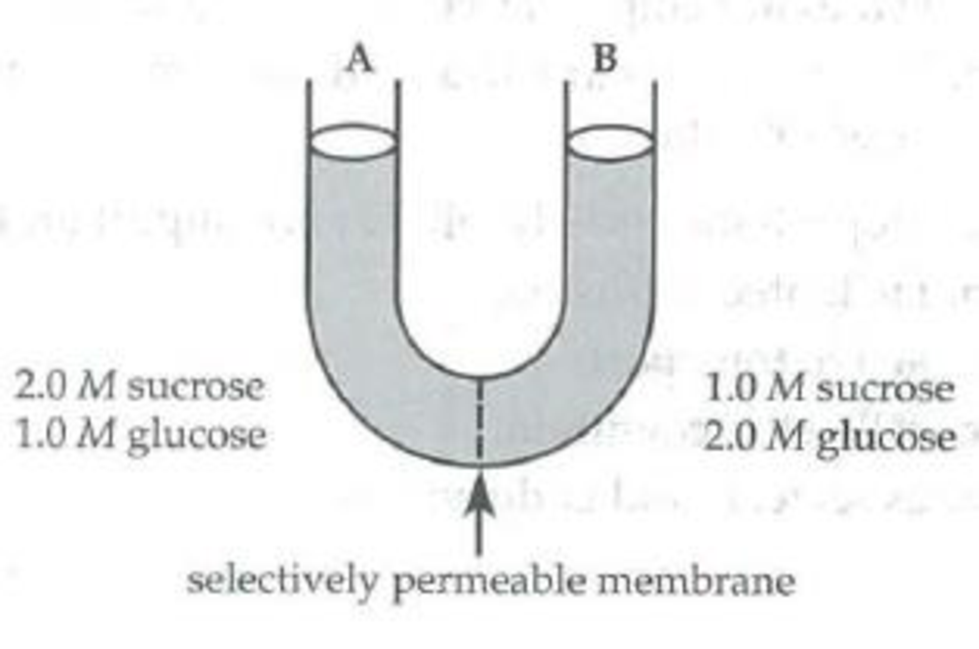
The solutions in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is filled with a solution of 2.0 M sucrose and 1.0 M glucose. Side B is filled with 1.0 M sucrose and 2.0 M glucose.
Initially, the solution in side A, with respect to that in side B,
- a. has a lower solute concentration.
- b. has a higher solute concentration.
- c. has an equal solute concentration.
- d. is higher in the tube.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
If a percent difference is a negative number, what does this tell us about the change in weight of the artificial cell? If the percent difference is a negative number, what does this tell us about the movement of water across the membrane?
The solutions in the two arms of the U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is filled with a solution of 2.0 M sucrose and 1.0 M glucose. Side B is filled with 1.0 M sucrose and 2.0 M glucose.
Initially, the solution in side A, with respect to that in side B, is what?
After the system reaches equilibrium, what changes are observed?
Define the following terms: a. semipermeable membrane b. hypotonic c. hypertonic d. crenation e. hemolysis
Chapter 7 Solutions
Study Guide for Campbell Biology
Ch. 7 - Label the components in the following diagram of a...Ch. 7 - a. Cite some experimental evidence that indicates...Ch. 7 - List the six major functions that membrane...Ch. 7 - What types of molecules have difficulty crossing...Ch. 7 - A solution of 1 M glucose is separated by a...Ch. 7 - a. What osmotic problems does the freshwater...Ch. 7 - Prob. 7IQCh. 7 - Prob. 8IQCh. 7 - a. How is cholesterol transported into human...Ch. 7 - Create a concept map to illustrate your...
Ch. 7 - The following diagram illustrates passive and...Ch. 7 - If a single layer of phospholipids coats the water...Ch. 7 - Glycoproteins and glycolipids are important for a....Ch. 7 - Prob. 3TYKCh. 7 - Prob. 4TYKCh. 7 - Prob. 5TYKCh. 7 - Use the following U-tube setup to answer questions...Ch. 7 - Use the following U-tube setup to answer questions...Ch. 7 - Use the following U-tube setup to answer questions...Ch. 7 - How much work is expended in diffusion? a. an...Ch. 7 - Prob. 10TYKCh. 7 - Prob. 11TYKCh. 7 - Water passes quickly through cell membranes...Ch. 7 - Facilitated diffusion of ions across a cellular...Ch. 7 - Prob. 14TYKCh. 7 - Which of the following describes cotransport? a....Ch. 7 - Prob. 16TYKCh. 7 - An animal cell moves potassium against its...Ch. 7 - Prob. 18TYKCh. 7 - Prob. 19TYKCh. 7 - In response to low blood pressure, the adrenal...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- A cell in placed in a beaker of water, and the cell shrinks. What can you infer about the relative concentrations of the solutes in the cell versus the beaker?arrow_forwardFor a lab on diffusion and osmosis, where a egg yolk was placed in a cup water the following question asks Consider a scenario in which the size of an egg yolk remains unchanged after in water soaking for an hour. What are two possible explanations as to why this occurs? What would two reasons be for the yolk to be unchanged after a hour in water?arrow_forwardWhy is it advantageous for the cell membrane to be fluid in nature?arrow_forward
- You have a beaker filled with a solution containing 2M glucose, 4M urea and 1M salt.Suspended in the solution is a cell that containing a solution of 1M glucose, 8M urea and 3Msalt. The membrane of the cell is permeable to glucose and salt but not urea. Answer each of thefollowing questions:a. Where will water move?b. Where will urea move?c. Where will glucose move?d. Where will salt move?e. What will happen to the volume of fluid inside the cell?f. What will happen to the osmolarity of the fluid inside the cell?arrow_forwardWhen a semipermeable sac filled with a solution containing 10% glucose is immersed in water, the fluid volume in the sac increases. What would happen if the sac solution was replaced with a 20% glucose solution?arrow_forwardWhich statement is NOT correct concerning the cell membrane? 1. The fatty acid hydrophobic "tails" are contained within what becomes a double- layered molecule. 2. This molecule provides an impermeable barrier for the cell from the watery outer environment. 3. The overall structure of the cell membrane results from the amphiphilic phospholipid molecules. 4. Because of the hydrophilic property of the phosphate containing "head", this "head* orients itself toward the "watery." cytoplasm and the extracellular fluid.arrow_forward
- Justify the parenteral use of hypotonic and hypertonic solutions? explain at your own wordsarrow_forwardThe beaker is divided into two compartments (C and D) that has equal volumes of solution separated by an artificial membrane that has a pore size of 23 Å (Angstrom). Explain the movement of solute after eight hours if comparment C has 17 % sucrose while the compartment D has 2 % sucrose (diameter of sucrose molecule = 9 Å (Angstrom))arrow_forwardWhen the potato cube is surrounded by a hypotonic solution what change in mass occurs in the potato cells?arrow_forward
- Give typing answer with explanation and conclusion Molecules move from higher to lower concentration through a transport protein in Group of answer choices simple diffusion. pinocytosis. facilitated diffusion. phagocytosis. Flag question:arrow_forwardCompare and contrast the following methods of a passing cell membrane in terms of movement with respect to the concentration gradient, use of ATP, and the use of transporters with examples. (Simple) Diffusion Facilitated Diffusion/Passive Transport Osmosis Active Transport Exocytosis Endocytosis (with its 3 subforms)arrow_forwardIn this part of the assignment, assume you have carried out the microscopic examination of the effects of osmosis in plant cells experiment The image in Figure 4 is of Elodea canadensis leaf cells in isotonic solution (normal saline). The image in Figure 5 is of Elodea canadensis leaf cells in 10% sucrose solution. The image in Figure 6 is of Elodea canadensis leaf cells in distilled water. a. Determine cell size and vacuole size. b. Calculate the percentage change in vacuole size for each treatment compared to the control. Use the formula: Experimental Size - Control Size/Control Size c. Based on these results, explain the tonicity of each treatment using experimental results to support your answer as would appear in the discussion section of a formal report (maximum four (4) lines). d. Was the percentage change in vacuole size for the experimental conditions relative to the control of similar magnitude? If the change was not of the same magnitude, explain the difference in effect…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning
Human Biology (MindTap Course List)BiologyISBN:9781305112100Author:Cecie Starr, Beverly McMillanPublisher:Cengage Learning Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning

Human Biology (MindTap Course List)
Biology
ISBN:9781305112100
Author:Cecie Starr, Beverly McMillan
Publisher:Cengage Learning

Concepts of Biology
Biology
ISBN:9781938168116
Author:Samantha Fowler, Rebecca Roush, James Wise
Publisher:OpenStax College
The Cell Membrane; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=AsffT7XIXbA;License: Standard youtube license