
Concept explainers
Interpretation:
The molecular geometry of
Concept introduction:
- Electron domain is the term used to indicate lone pair and bond pair of electrons.
- Lewis structures is also known as Lewis dot structures which represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
- In VSEPR, the geometry of the molecule is explained based on minimizing electrostatic repulsion between the molecules’ valence electrons around a central atom
- Electro-domain geometry includes both bond pairs and lone pairs of central atom for determining the geometry of molecule.
- Molecular geometry includes only the bond pairs of central atom for determining the geometry of the molecule.
Correct answer:
Trigonal pyramidal
Explanation of Solution
Reason for correct option
To determine: the electron-domain geometry and molecular geometry of
Trigonal pyramidal
In
Three bromine atoms are bonded to phosphorus atom. So phosphorus is the central atom in this molecule.
A Lewis structure represents the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule.
Therefore,
The Lewis structure of
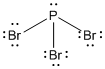
According to VSEPR theory, the geometry of the molecule is explained based on minimizing electrostatic repulsion between the molecules’ valence electrons around a central atom
The Lewis structure of
Electro-domain geometry includes both bond pairs and lone pairs of central atom for determining the geometry of molecule.
Therefore,
The electron-domain geometry of
Molecular geometry includes only the bond pairs of central atom for determining the geometry of the molecule.
Therefore,
The molecular geometry of
Hence the correct option for molecular geometry of
Reasons for incorrect options:
Writes all the molecular geometry of
(a) Trigonal planar
(b) Tetrahedral
(d) Bent
(e) T-shaped
The molecular geometry of
So, this molecular geometry of
Hence the incorrect options are a, b, d and e.
Conclusion
The correct optionfor molecular geometry of
Want to see more full solutions like this?
Chapter 7 Solutions
CHEM: ATOM FIRST V.2 W/ACCESS >C<
- Sketch the resonance structures for the N2O molecule. Is the hybridization of the N atoms the same or different in each structure? Describe the orbitals involved in bond formation by the central N atom.arrow_forwardWhat is the hybridization of the central atom in each of the following? (a) BeH2 (b) SF6 (c) PO43 (d) PCl5arrow_forwardWhat is the NO bond order in nitrogen monoxide, NO? (a) 2 (b) 2.5 (c) 3.0 (d) 1arrow_forward
- What is the hybridization of the central atom in the tetrafluoroiodide IF−4 anion?arrow_forwardWhat nonmetal in Period 3 forms a single bond and one triple bond?arrow_forwardQ. Hybridization is an energy requiring process, yet it occurs, why? Discuss shape of the following: [Cu (NH3)4] + and [Ni (CN)4]-2arrow_forward
- Consider the molecule PF4Cl. (a) Draw a Lewis structure forthe molecule, and predict its electron-domain geometry.(b) Which would you expect to take up more space, a P¬Fbond or a P¬Cl bond? Explain. (c) Predict the molecular geometryof PF4Cl. How did your answer for part (b) influenceyour answer here in part (c)? (d) Would you expect the moleculeto distort from its ideal electron-domain geometry? Ifso, how would it distort?arrow_forward1. What is the hybridization for each of the indicated atoms in the following compound? 2. Which is not an acceptable Lewis structure for the anion (CH2NCO)–?arrow_forwardWhat is the hybridization of As in AsCl3?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





