
Concept explainers
(a)
Interpretation:
The difficulty of that would come on models of the cis and trans isomers of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings, and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The cis isomers of
Explanation of Solution
The structure of cis and trans isomers of
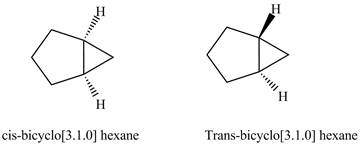
Figure 1
The formation of trans isomers of
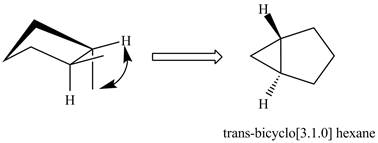
Figure 2
In the formation of trans isomers of
The formation of cis isomers of
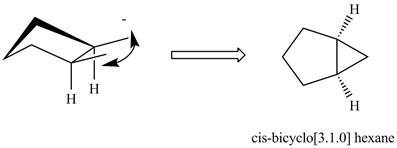
Figure 3
In the formation of cis isomers of
It is easier to form a three-membered ring by the methyl at the cis position and the methyl groups at trans position. Therefore, cis isomers of
(b)
Interpretation:
The difficulty of that would come on models of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The compound
Explanation of Solution
The structure of
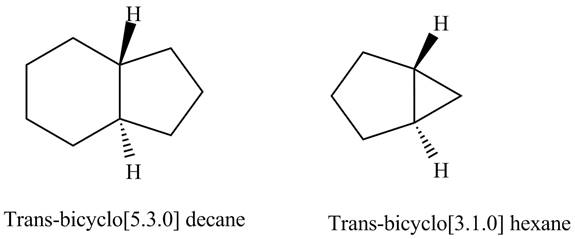
Figure 4
The formation of
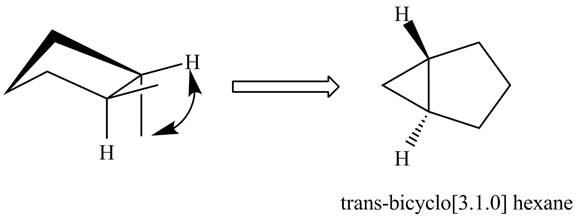
Figure 2
In the formation of
The formation of
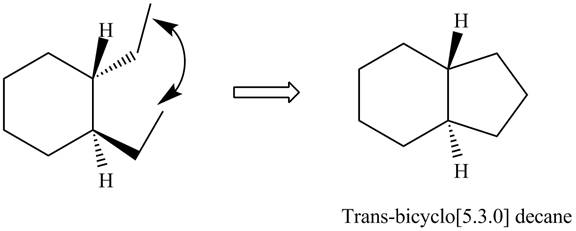
Figure 5
In the formation of
The methyl groups in case of
Want to see more full solutions like this?
Chapter 7 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- The potential energy of a CH3 group in ethane as it is rotated around the C-C bond can be written V= 1/2V0(1 +cos φ), where φ is the azimuthal angle as shown and V0 = 11.6 kJ mol-1. (a) What is the change in potential energy between the trans and fully eclipsed conformations? (b) Show that for smal lvariations in angle, the torsional (twisting) motion around the C-C bond can be expected to be that of a harmonic oscillator. (d) Estimate the vibrational frequency of this torsional oscil lation.arrow_forwardPerform a conformational analysis of 1-bromo-2-chloroethane, BrCH2CH2Cl. Pay attention to the relative energies of the various conformations, but do not concern yourself with the actual energy values.arrow_forwardUse bond-dissociation enthalpies to calculate the AH° for each of the following reactions. CH3CH2CH3+H2 → CH3CH3+CH4 CH3CH2CL+HI → CH3CH2I+HCIarrow_forward
- Which will be more stable, cis or trans-1,4-tert-butylcyclohexane? Explain by drawing their structures?arrow_forwardWhy is the percentage of molecules with the substituent in an equatorial position greater for isopropylcyclohexane than for fluorocyclohexane?arrow_forwardThe German chemist Wilhelm Kӧrner (1839-1925) observed in 1974 that each of the three isomers of dibromobenzene, A, B, and C, gave a different number of tribromobenzenes upon further bromination, allowing him to assign their respective structures. Try to do the same and assign structures to A, B, and C based on the following results: A gives two tribromobenzenes in comparable amounts B gives three tribromobenzenes, one of them in minor quantities C gives only one tribromobenzenearrow_forward
- Between the cis and trans configuration of 1-chloro-3-methylcyclohexane, which is the most stable stereoisomer and configuration? To prove your argument, use your models or a modelling software to model cis- and trans-1-chloro-3-methylcyclohexane (show all the hydrogens and ensure proper chair conformation) and use arrows to show where steric strain occurs. Include these photos in your answer.arrow_forwardDraw the structure of the organic product of the reaction between cyclohexene and H2SO4, H2O. Use the wedge/hash bond tools to indicate stereochemistry where it exists. Separate multiple products using the + sign In cases where there is more than one answer, just draw one. Please make your answer clear. I did not understand the first answer because it was hand -writtenarrow_forwardHow many degrees of unsaturation (double bond equivalents) does C4H10ClNO have?arrow_forward
- 3. Which of the following compounds would you expect to have the greatest density: hexane,1-bromohexane, 1-fluorohexane, or 1-chlorohexane? Explain your answer.arrow_forwardtrans-1,4-dimethylcyclohexane has two conformations with a difference in energy of 3.6 kcal/mol. What is the cause of this difference? Hint: this is twice the difference in methylcyclohexane!arrow_forwardRank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Clarrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

