
Concept explainers
(a)
Interpretation:
The structure of all products formed in the given reaction is to be shown.
Concept Introduction:
The compounds which have same molecular formula but different connectivity of atoms are known as constitutional isomers. Chiral compounds are those compounds which contain an asymmetric carbon atom. Chiral molecules are optically active molecules. Stereocentre can be an atom, bond, or any point in molecule at which interchange of two groups form a stereoisomer.
Answer:
(1) The structures of all products formed in the given reactions are shown below.
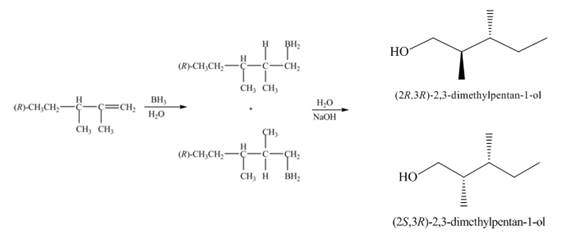
(2) The structures of all products formed in the given reactions are shown below.
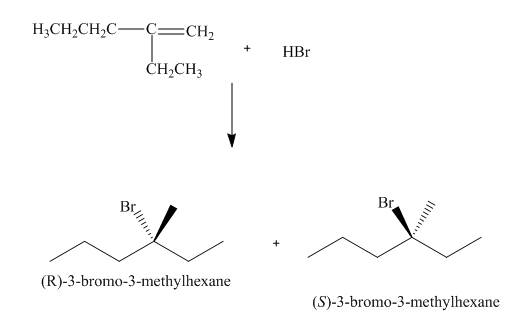
(3) The structures of all products formed in the given reactions are shown below.
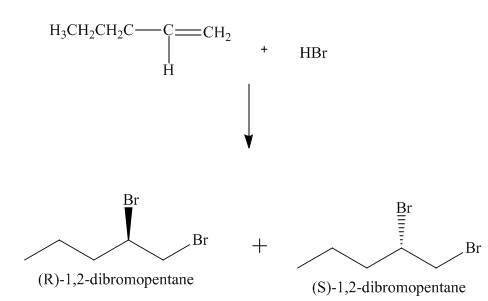
(4) The structures of all products formed in the given reactions are shown below.
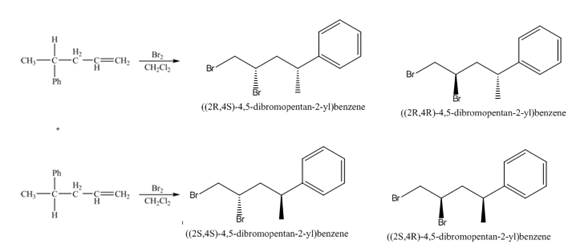
(5) The structures of all products formed in the given reactions are shown below.
(6) The structures of all products formed in the given reactions are shown below.
Explanation:
(1) The
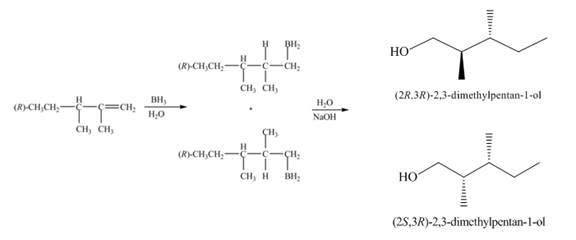
Figure 1
(2) The electrophilic addition of hydrogen bromide takes place on the carbon-carbon double bond of the alkene. The addition follows markonikov’s rule, the hydrogen atom goes to the less substituted carbon atom. The products formed in the given reaction are shown below.

Figure 2
(3) The addition reaction of bromine on the alkene takes place. The addition of bromine on alkene is anti addition. The cyclic bromonium ion intermediate is formed in the reaction. The product formed in the given reaction is shown below.

Figure 3
(4) The addition reaction of bromine on the alkene takes place. The addition of bromine on alkene is anti addition. The cyclic bromonium ion intermediate is formed in the reaction. The product formed in the given reaction is shown below.
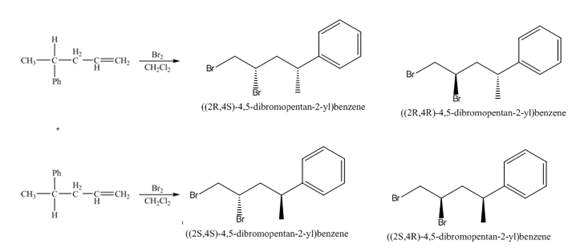
Figure 4
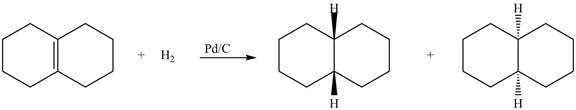
(5) In the reduction reaction of alkene in presence of palladium metal the hydrogen gets adsorbed on the metal surface. Alkene reduction to form
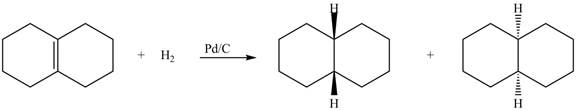
Figure 5
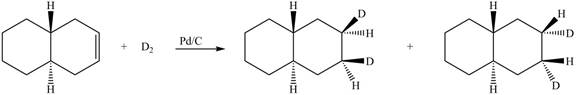
(6) In the reduction reaction of alkene in presence of palladium metal,
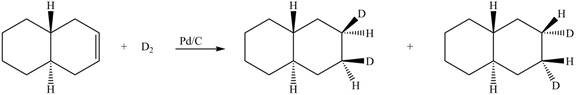
Figure 6
Conclusion:
The products formed in the given reactions are shown in Figure 1, Figure 2, Figure 3, Figure 4, Figure 5 and Figure 6.
Answer to Problem 7.42AP
(1) The structures of all products formed in the given reactions are shown below.

(2) The structures of all products formed in the given reactions are shown below.

(3) The structures of all products formed in the given reactions are shown below.

(4) The structures of all products formed in the given reactions are shown below.

(5) The structures of all products formed in the given reactions are shown below.

(6) The structures of all products formed in the given reactions are shown below.

Explanation of Solution
(1) The alkene undergoes hydroboration-oxidation reaction to form alcohol compounds. The borane gets added to the double bond. Then it will undergo oxidation reaction to form the alcohol compound. The products formed in the given reaction are shown below.

Figure 1
(2) The electrophilic addition of hydrogen bromide takes place on the carbon-carbon double bond of the alkene. The addition follows markonikov’s rule, the hydrogen atom goes to the less substituted carbon atom. The products formed in the given reaction are shown below.

Figure 2
(3) The addition reaction of bromine on the alkene takes place. The addition of bromine on alkene is anti addition. The cyclic bromonium ion intermediate is formed in the reaction. The product formed in the given reaction is shown below.

Figure 3
(4) The addition reaction of bromine on the alkene takes place. The addition of bromine on alkene is anti addition. The cyclic bromonium ion intermediate is formed in the reaction. The product formed in the given reaction is shown below.

Figure 4
(5) In the reduction reaction of alkene in presence of palladium metal the hydrogen gets adsorbed on the metal surface. Alkene reduction to form alkane takes place forming syn addition product. The products formed in the given reaction are shown below.

Figure 5
(6) In the reduction reaction of alkene in presence of palladium metal,

Figure 6
The products formed in the given reactions are shown in Figure 1, Figure 2, Figure 3, Figure 4, Figure 5 and Figure 6.
(b)
Interpretation:
The stereochemical relationship between products formed is to be stated.
Concept Introduction:
Stereoisomers which are non-superimposable and not mirror image are known as diastereomer. The compound must contain two or more than two stereocentre. Diastereomer are non identical stereoisomers. The pair of stereoisomer which are mirror image of each other are known as enantiomers. Enantiomers are non-congruent mirror images. If the molecules are placed on top of each other they will not give same molecule.
Answer to Problem 7.42AP
(1) The products formed are diastereomers.
(2) The products formed are enantiomers.
(3) The products formed are enantiomers.
(4) The two pairs of products formed are diastereomers.
(5) Only one product is formed.
(6) Only one product is formed.
Explanation of Solution
(1) The products formed are non-congruent and not mirror image of each other. Hence they are diastereomers.
(2) The products formed are non-congruent mirror image of each other. Therefore, they are enantiomers.
(3) The products formed are non-congruent mirror image of each other. Therefore, they are enantiomers.
(4) The products formed are non-congruent and not mirror image of each other. Hence they are diastereomers.
(5) Only one product is formed.
(6) Only one product is formed.
The stereochemical relationship between products formed in reaction (1) and (4) are diastereomers and reaction (2) and (3) are enantiomers.
(c)
Interpretation:
The products are formed in identical or different amount is to be stated.
Concept Introduction:
The electrophilic addition reaction on the alkene can takes place from any side of alkene. The electrophile can attack the carbon double bond from above or below the plane. The probability of attack from both sides is equal.
Answer to Problem 7.42AP
(1) The products are formed in equal amount.
(2) The products are formed in equal amount.
(3) The products are formed in equal amount.
(4) The products are formed in equal amount.
(5) Only one product is formed.
(6) Only one product is formed.
Explanation of Solution
(1) The alkene is a planar molecule. The reaction on the carbon-carbon double bond of alkene can takes place from above or below the plane. Therefore, products are formed in equal amount.
(2) The alkene is a planar molecule. The reaction on the carbon-carbon double bond of alkene can takes place from above or below the plane. Therefore, products are formed in equal amount.
(3) The alkene is a planar molecule. The reaction on the carbon-carbon double bond of alkene can takes place from above or below the plane. Therefore, products are formed in equal amount.
(4) The alkene is a planar molecule. The reaction on the carbon-carbon double bond of alkene can takes place from above or below the plane. Therefore, products are formed in equal amount.
(5) Only one product is formed.
(6) Only one product is formed.
The products are formed in equal amount in reaction (1), (2), (3), (4) and only one product is formed in reaction (5) and (6).
(d)
Interpretation:
The products which have different physical properties is to be stated.
Concept Introduction:
Stereoisomers which are non-superimposable and not mirror image are known as diastereomer. The compound must contain two or more than two stereocentre. Diastereomer are non identical stereoisomers. The pair of stereoisomer which are mirror image of each other are known as enantiomers. Enantiomers are non-congruent mirror images. Diastereomers shows different physical properties and enantiomers show same physical properties.
Answer to Problem 7.42AP
(1) The products formed in the given reaction will have different physical properties.
(2) The products formed in the given reaction will have identical physical properties.
(3) The products formed in the given reaction will have identical physical properties.
(4) The products formed in the given reaction will have different physical properties.
(5) Only one product is formed.
(6) Only one product is formed.
Explanation of Solution
(1) The products formed in the given reaction are diastereomer. Diastereomers show different physical properties. Therefore, the products formed in the given reaction will have different physical properties.
(2) The products formed in the given reaction are enantiomers. Enantiomers show identical physical properties. Therefore, the products formed in the given reaction will have identical physical properties.
(3) The products formed in the given reaction are enantiomers. Enantiomers show identical physical properties. Therefore, the products formed in the given reaction will have identical physical properties.
(4) The products formed in the given reaction are diastereomer. Diastereomers show different physical properties. Therefore, the products formed in the given reaction will have different physical properties.
(5) Only one product is formed.
(6) Only one product is formed.
The products formed in the reaction (1) and reaction (4) will have different physical properties. The products formed in the reaction (2) and reaction (3) will have identical physical properties. In reaction (5) and (6) only one product is formed.
Want to see more full solutions like this?
Chapter 7 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- Provide the complete arrow pushing mechanism and provide the product for the reactions. Be sure to consider stereochemistry in your product(s). Give a short concise statement about the step that determines the stereoselectivity, or lack of, for each reaction. Also give a statementarrow_forwardFor the following, complete the reactions with the predominant product or products. You must indicate stereochemistry with wedges and dashes. If a racemic mixture is created, you must write "racemic" under the structures.arrow_forwardProvide a reasonable stepwise mechanism for the following transformation. Show the structure of any intermediates and use the arrow formalism to account for electron flow. Draw the structure of the regioisomer that might form and suggest a reason why it would not be produced.arrow_forward
- Give the structure of the product and/or intermediates of the following reactions. Indicate, where appropriate, both regiochemistry and stereochemistry.arrow_forwardSince compounds A and B are in different comformations, how do the products of the elimation reaction differ in conformation between these two compounds? Please provide visual drawing to explain.arrow_forwardProvide mechanisms that explain the following results. Be sure to show any intermediates that are formed, and indicate the flow of electrons with arrows. Indicate whether or not enantiomers are formed or a single stereoisomer for each product.arrow_forward
- Draw the structure of the product, substrate, or condition in the following reactions. Clearly show the stereochemistry.arrow_forwardA. Arrange the following radicals in order of decreasing rate of bromination. Justify your answer. B. Trehalose and isomaltose are both dimers of glucose. However, they have considerablydifferent reactivities. Concisely explain why these differences are observed. -Isomaltose is a reducing sugar while trehalose is not.-Trehalose is very resistant to acid hydrolysis while isomaltose can be acid-hydrolyzed withease.arrow_forwardIdentify compound R, indicating the relative stereochemistry whilst providing a curved arrow mechanism to account for its formation. can you also explain how you decide the correct stereochemistry from the basics thank youarrow_forward
- Give the stereochemical outcome for each of the following reactions and assign the stereochemical relationship between the products:arrow_forwardRepresent the products that would be obtained in each of the reactions below (leaving the stereochemistry clearlyrepresented, in the case of products from stereospecific reactions) and classify them in relation to what occurswith the substrate, indicating whether they are: mild oxidation, oxidative cleavage or reduction.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





