
Concept explainers
(a)
Interpretation:
The difficulty of that would come on models of the cis and trans isomers of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings, and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The cis isomers of
Explanation of Solution
The structure of cis and trans isomers of
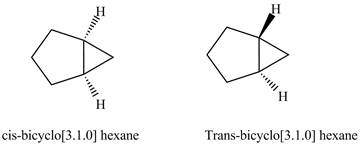
Figure 1
The formation of trans isomers of
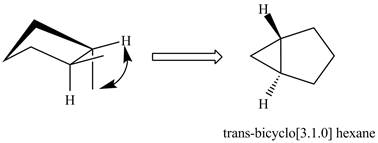
Figure 2
In the formation of trans isomers of
The formation of cis isomers of
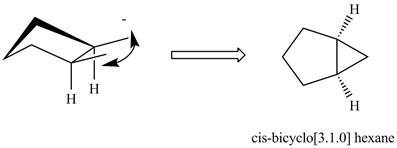
Figure 3
In the formation of cis isomers of
It is easier to form a three-membered ring by the methyl at the cis position and the methyl groups at trans position. Therefore, cis isomers of
(b)
Interpretation:
The difficulty of that would come on models of
Concept introduction:
The organic compounds which consist of two cyclic rings fused together are known as bicyclic compounds. Fused rings, bridged rings and spiro rings are the examples of bicyclic compounds. The stability of bicyclic compounds depends on the ring strain.
Answer to Problem 7.21P
The compound
Explanation of Solution
The structure of
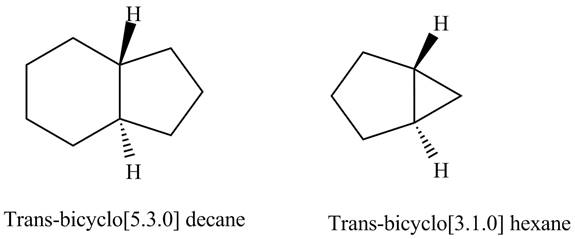
Figure 4
The formation of
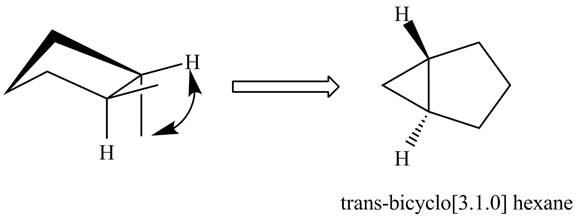
Figure 2
In the formation of
The formation of
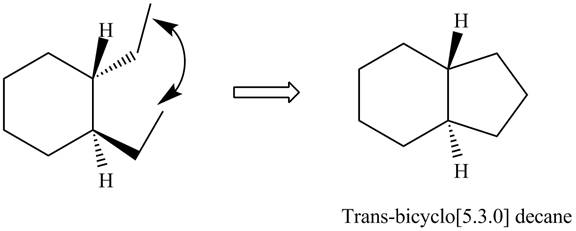
Figure 5
In the formation of
The methyl groups in case of
Want to see more full solutions like this?
Chapter 7 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- (a) (b) 1. CH3MgBr 2. H3O+ H3C OH I H3C-C-C-CH3 H3C H ОН H CH3 CH3 H3C CH3 H3C H₂SO4 C=Carrow_forward1. Which of the following formulae represent the same compound? (1) CH;CH,CH;CHCH3 (3) CH-CНCH, 1. CH2 (2) CH3 CH3 CHCH;CH2 CH3 CH3 CH;CH3 (4) CH;CHCH,CH,CH3 (5) CH;CH,CHCH;CH3 CH3 CH3 A. (2) and (3) only В. (1), (2) and (4) only С. (1), (4) and (5) only D. (1), (2), (3) and (4) onlyarrow_forwardnumber 3arrow_forward
- Using solid and dashed wedges, draw the structure of (1S, 2S, 4R, 5R)-1,5-dibromo-2,4- dichlorocyclohexane, then draw the most stable conformer.arrow_forwardThe enthalpy for hydrogenation of different isomers of butene is shown in the following figure. Which statement below best interprets this figure? Hy I3D H2 7k mot 5kJ mot + H2 AH-120 kJ mot AH"-127 kJ mot AH-115 kJ mot © GMU 2020 Stability and hydrogenation enthalpies are not correlated. Butene II is less stable than butene I by 7 kJ/mol. O Butene Il is 7 kJ/mol more stable than I and 5 kJ/mol more stable than III. Butene II is 5 kJ/mol less atable than butene IlI. Enthalpyarrow_forwardPlease don't provide handwritten solution ...arrow_forward
- Font Paragraph Predict the main organic product of the following reactions: (a) CH2-CHCHCH3 H*, H2O, heat CH3 (b) CH-СНСНСH3 Br2 CH3 (c) CH-CHCHСH3 (1) BH3 CH3 (2) H2О2, ОН-arrow_forwardGive the relationships between the following pairs of structures (a), (c), (d), (e). The possible relationships are: same compound, constitutional isomer, cis-trans isomers, not isomers (different molecular formula). 2-35 Give the relation isomers, constitutional (structural) isomers, (a) CH₂CH₂CH₂CH3 and (CH3)3CH (c) (e) CH CH CH₁ and CH₂ and CH₂ CH, (d) CH3 CH₂ CH₂ CH₂-CH₂ and CH₂CH3 CH3 C=CH₂ CH3 and CH3. CH₂-CHarrow_forwardFrom the data in Figure 4-12 and Table 4-1, estimate the percentages of molecules that have their substituents in an axial orientation for the following compounds: (a) Isopropylcyclohexane (b) Fluorocyclohexane (c) Cyclohexanecarbonitrile, C6H11CNarrow_forward
- Increased substitution around a bond leads to increased strain. Take the four substituted butanes listed below, for example. For each compound, sight along the C2-C3 bond and draw Newman projections of the most stable and least stable conformations. Use the data in Table 3-5 to assign strain-energy values to each conformation. Which of the eight conformations is most strained? Which is least strained? (a) 2-Methylbutane (b) 2,2-Dimethylbutane (c) 2,3-Dimethylbutane (d) 2,2,3-Trimethylbutanearrow_forward(1) C (CH ),CO K (CH2),COH Сн,о- Д НО Aarrow_forwardSet C. Some Relatively Easy Molecules (1) SF;CI (2) Chlorobenzene (3) 1,2-Dichlorobenzene (4) 1,3-Dichlorobenzene (5) 1,4-Dichlorobenzene (6) 1,3,5-Trichlorobenzene (7) 1,2,3-Trichlorobenzene (8) 1,2,4-Trichlorobenzene (9) trans-(CrCl,(H,O).J* (ignore H atoms) (10) (11) OPCI, (12) trans-Pt(NH,),Cl; (ignore H atoms) (13) cis-Pt(NH,),Cl, (ignore H atoms) (14) BICIF (15) What is the point group for each of the following substituted cyclo- butanes? Assume that C,H, itself has D symmetry and that replacing an H by X or Y changes no other structure parameters. (a) (d)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

