
Concept explainers
(a)
Interpretation:
Whether the product of the given step can eliminate a leaving group to form a different compound than the reactant is to be predicted. The product for the given nucleophilic elimination step with appropriate curved arrows is to be drawn.
Concept introduction:
In the nucleophilic elimination step, the more electronegative atom bears full or partial negative charge. This is an electron rich atom, and the less electronegative atom is relatively electron poor. The curved arrow drawn from the lone pair of electron rich atom points to the bonding region between the more electronegative atom and less electronegative atom representing the electron flow from the electron rich site to the electron poor site. The second curved arrow is drawn to represent the breaking of the bond between the less electronegative atom and leaving group to avoid exceeding an octet on the less electronegative atom.
Answer to Problem 7.27P
Products formed after the elimination of the leaving group are not the same as the reactant. Product formed in the nucleophilic elimination step with appropriate curved arrows is drawn as:
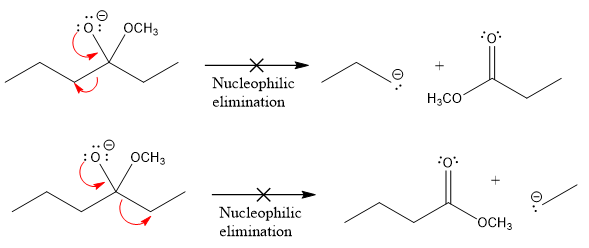
Explanation of Solution
Product for the given nucleophilic addition step is:
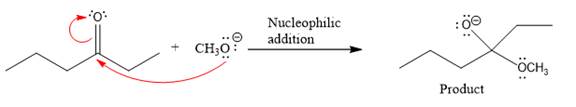
In the given product, there are two possible groups that can leave to form two different products.
In the first nucleophilic elimination step, the oxygen atom with negative charge is an electron rich site, and the carbon bonded to it is an electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:
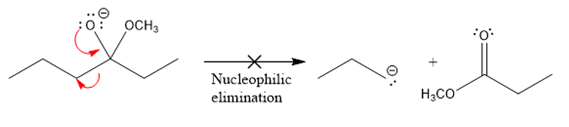
The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents that this nucleophilic elimination is unfeasible as
In the second nucleophilic elimination step, the oxygen atom with negative charge is the electron rich site, and the carbon bonded to it is the electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:
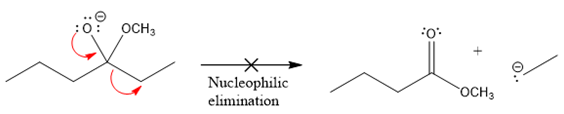
The first curve arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents that this nucleophilic elimination is unfeasible as
Products formed in the elimination steps are different from the reactant in the given nucleophilic addition step.
(b)
Interpretation:
Whether the product of the given step can eliminate a leaving group to form a different compound than the reactant is to be predicted. The product for the given nucleophilic elimination step with appropriate curved arrows is to be drawn.
Concept introduction:
In the nucleophilic elimination step, the more electronegative atom bears full negative charge or partial negative charge. This is the electron rich atom and the less electronegative atom is relatively electron poor. The curved arrow drawn from the lone pair of electron rich atom points to the bonding region between the more electronegative atom and less electronegative atom representing the electron flow from the electron rich site to the electron poor site. The second curved arrow is drawn to represent the breaking of bond between the less electronegative atom and leaving group to avoid exceeding an octet on the less electronegative atom.
Answer to Problem 7.27P
The product formed after the elimination of the leaving group is not the same as the reactant. Product formed in the nucleophilic elimination step with an appropriate curved arrow is drawn as:
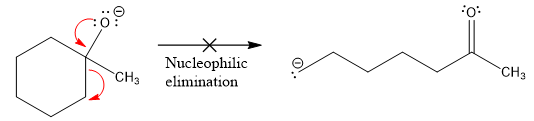
Explanation of Solution
Product for the given nucleophilic addition step is:
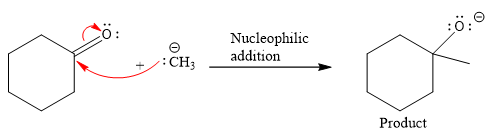
In the nucleophilic elimination step, the oxygen atom with negative charge is an electron rich site, and the carbon bonded to it is an electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:

The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents this nucleophilic elimination is unfeasible as
Product formed in the elimination step is different from the reactant in the given nucleophilic addition step.
(c)
Interpretation:
The product of the given step can eliminate a leaving group to form different compound than reactant is to be predicted. The product for the given nucleophilic elimination step with appropriate curved arrows is to be drawn.
Concept introduction:
In nucleophilic elimination step, the more electronegative atom bears full negative charge or partial negative charge. This is the electron rich atom and the less electronegative atom is relatively electron poor. The curved arrow drawn from the lone pair of electron rich atom points to the bonding region between the more electronegative atom and less electronegative atom representing the electron flow from electron rich site to electron poor site. The second curved arrow drawn to represent the breaking of bond between the less electronegative atom and leaving group to avoid exceeding an octet on the less electronegative atom.
Answer to Problem 7.27P
The products formed after the elimination of the leaving group are not the same as the reactant. Product formed in the nucleophilic elimination step with appropriate curved arrow is drawn as:
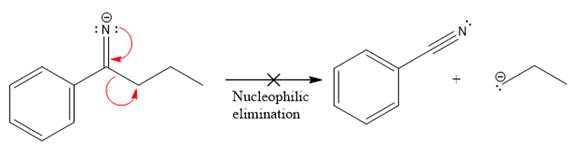
Explanation of Solution
Product for the given nucleophilic addition step is:
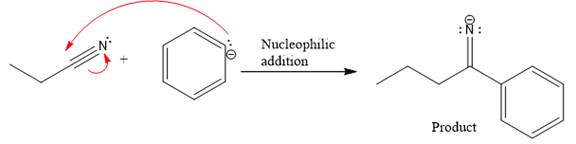
In the nucleophilic elimination step, the nitrogen atom with negative charge is electron rich site, and the carbon bonded to it is electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:

The first curved arrow is drawn from the lone pair of negatively charged nitrogen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents this nucleophilic elimination is unfeasible as
Product formed in the elimination step is different from the reactant in the given nucleophilic addition step.
(d)
Interpretation:
Whether the product of the given step can eliminate a leaving group to form different compound than reactant is to be predicted. The product for the given nucleophilic elimination step with appropriate curved arrows is to be drawn.
Concept introduction:
In nucleophilic elimination step, the more electronegative atom bears full negative charge or partial negative charge. This is the electron rich atom and the less electronegative atom is relatively electron poor. The curved arrow drawn from the lone pair of electron rich atom points to the bonding region between the more electronegative atom and less electronegative atom representing the electron flow from electron rich site to electron poor site. The second curved arrow is drawn to represent the breaking of bond between the less electronegative atom and leaving group to avoid exceeding an octet on the less electronegative atom.
Answer to Problem 7.27P
Products formed after the elimination of the leaving group are not the same as the reactant. Product formed in the nucleophilic elimination step with appropriate curved arrow is drawn as:
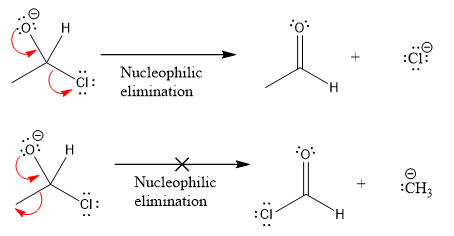
Explanation of Solution
Product for the given nucleophilic addition step is:
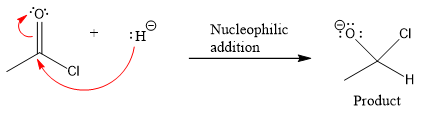
In the first nucleophilic elimination step, the oxygen atom with negative charge is electron rich site, and the chlorine atom is a good leaving group. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:
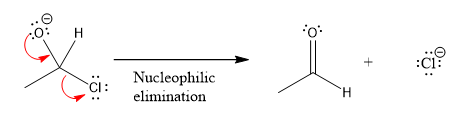
The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step.
In the second nucleophilic elimination step, the oxygen atom with negative charge is electron rich site and the carbon bonded to it is electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:
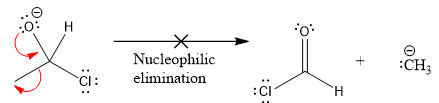
The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents this nucleophilic elimination is unfeasible as
Products formed in the elimination steps are different from the reactant in the given nucleophilic addition step.
(e)
Interpretation:
Whether the product of the given step can eliminate a leaving group to form different compound than reactant is to be predicted. The product for the given nucleophilic elimination step with appropriate curved arrows is to be drawn.
Concept introduction:
In nucleophilic elimination step, the more electronegative atom bears full negative charge or partial negative charge. This is the electron rich atom and the less electronegative atom is relatively electron poor. The curved arrow is drawn from the lone pair of electron rich atom points to the bonding region between the more electronegative atom and less electronegative atom representing the electron flow from electron rich site to electron poor site. The second curved arrow is drawn to represent the breaking of bond between the less electronegative atom and leaving group to avoid exceeding an octet on the less electronegative atom.
Answer to Problem 7.27P
Products formed after the elimination of the leaving group are not same as the reactant. Product formed in the nucleophilic elimination step with appropriate curved arrow is drawn as:
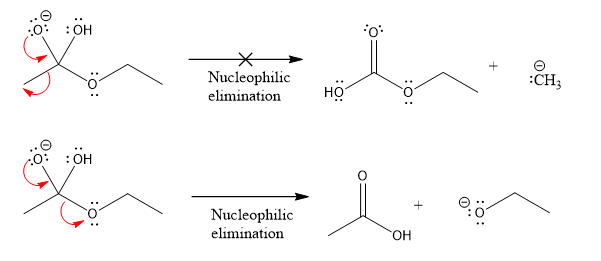
Explanation of Solution
Product for the given nucleophilic addition step is:
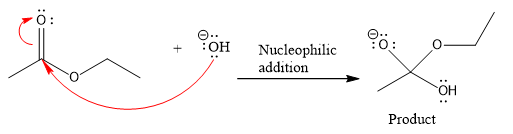
In the given product, there are two possible groups that can leave to form two different products.
In the first nucleophilic elimination step, the oxygen atom with negative charge is electron rich site, and the carbon bonded to it is electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:
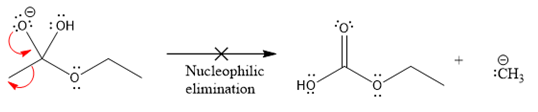
The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents this nucleophilic elimination is unfeasible as
In the second nucleophilic elimination step, the oxygen atom with negative charge is electron rich site and
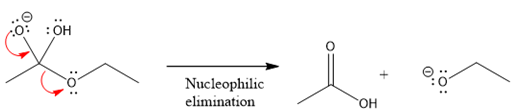
The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step.
Products formed in the elimination steps are different from the reactant in the given nucleophilic addition step.
(f)
Interpretation:
Whether the product of the given step can eliminate a leaving group to form different compound than reactant is to be predicted. The product for the given nucleophilic elimination step with appropriate curved arrows is to be drawn.
Concept introduction:
In nucleophilic elimination step, the more electronegative atom bears full negative charge or partial negative charge. This is the electron rich atom and the less electronegative atom is relatively electron poor. The curved arrow drawn from the lone pair of electron rich atom points to bonding region between the more electronegative atom and less electronegative atom representing the electron flow from electron rich site to electron poor site. The second curved arrow is drawn to represent the breaking of bond between the less electronegative atom and leaving group to avoid exceeding an octet on the less electronegative atom.
Answer to Problem 7.27P
Products formed after the elimination of the leaving group are not same as the reactant. Product formed in the nucleophilic elimination step with appropriate curved arrow is drawn as:
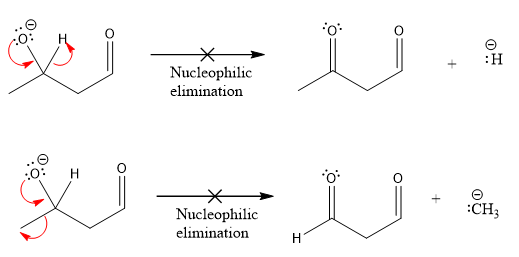
Explanation of Solution
Product for the given nucleophilic addition step is:

In the given product, there are two possible groups that can leave to form two different products.
In the first nucleophilic elimination step, the oxygen atom with negative charge is electron rich site and the carbon bonded to it is electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:

The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents this nucleophilic elimination is unfeasible as
The second nucleophilic elimination step, the oxygen atom with negative charge is electron rich site and the carbon bonded to it is electron poor site. The curved arrow mechanism for this given nucleophilic elimination step forming the respective product is:

The first curved arrow is drawn from the lone pair of negatively charged oxygen to the mid of
The respective product formed is different from the reactant in the given nucleophilic addition step. The X sign on the arrow represents this nucleophilic elimination is unfeasible as
Products formed in the elimination steps are different from the reactant in the given nucleophilic addition step.
Want to see more full solutions like this?
Chapter 7 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- What best defines a transition state?arrow_forwardFor each of the following elimination reactions, draw the major product and the minor product (if there is one). E 10. 11. 12. Br Br Br KOH H₂O KOH DMF KO'Bu DMFarrow_forwardExplain how you can tell from the energy diagram that the reaction with the catalyst in Fig. 8.4 isfaster than the reaction without the catalyst.arrow_forward
- Identify the electrophile and the nucleophile in each of the following reaction steps. Then draw curved arrows to illustrate the bond-making and bond-breaking processes.arrow_forward7. (Chapters 6 and 8) Within the following set, which is more stable, and why? CH3 CH3 H3C- -C=CH- CH2 H2C=Ć- -CH CH3 8. (Chapter 12) What type of instability will an intermediate need to address following the reaction of a nucleophile/base that has a negative charge with a pi bond that has uneven electron distribution between atoms with different electronegativities (C=O)? 9. (Chapter 9) Circle the carbon that will be unstable in the intermediate of the following reaction. Then, state the reason for your choice, and also indicate what type of instability it will be. H,C-CH,- C ECH with NaNH2 10. (Chapters 12 and 13) What are three sources used to provide electrons to an electron-deficient carbon with a leaving group? 1. 2. 3.arrow_forwardPlace the appropriate solvent (DMSO or HOCH3) under the reaction arrow for each of the following substitution reactions substitution reactions, so that they proceed in the forward direction. Explain your choice at the right. 6. .CI Nal Solvent? NaCI Solvent?arrow_forward
- 1. What is the nucleophile in this reaction? 2. Draw the energy profile for this reaction.arrow_forwardDraw a complete, step-wise, curved arrow mechanism for each reaction shown below. You don't need to worry about stereochemistry for these problems. It may help if you take the following steps. 1) Find the nucleophile and the electrophile. 2) Determine the major functional group present in the nucleophile and electrophile. 3) Determine the type of reaction this particular nucleophile/electrophile pair is likely to participate in 4) Draw the mechanism that corresponds with this reaction type. a) OH cat. H2SO4 HO Cl2 b) :OHarrow_forwardDraw the major product for the oxidation reaction. Then, draw the proper FULL electron-pushing mechanism for the reaction, including ALL intermediates (with formal charges) and electron-pushing arrows. Label the electrophile and nucleophile in each step.arrow_forward
- Draw a reaction coordinate diagram for the reaction. (Hint: An alkyl halide is more stable than an alkene.) Draw the structure of the intermediate/s and their location/s in the above reaction coordinate diagram.arrow_forward1. Copy and complete the following table by indicating whether each of the following scenarios would either INCREASE or DECREASE the rate of reaction. Scenario Putting reaction vessel in the fridge Decreasing temperature Removing source of heat energyN Lowering temperature Putting reaction mixture in a hot oven Rate of Reactionarrow_forwardConsider this nucleophilic substitution reaction. 1. Highlight the electrophilic carbon in red, and highlight the leaving group in blue. Highlight the atom in the nucleophile that will attack the electrophilic center in green. Only atoms need to be highlighted and not the lone pairs or formal charges. 2. Draw the product(s) of the reaction. Include all lone pairs.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
