
Concept explainers
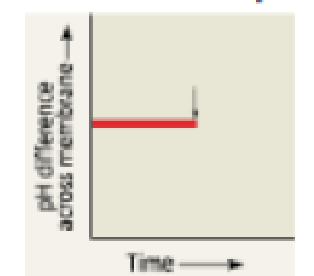
DRAW IT The graph here shows the pH difference across the inner mitochondrial membrane over time in an actively respiring cell. At the time indicated by the vertical arrow, a
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Campbell Biology in Focus (2nd Edition)
Additional Science Textbook Solutions
Laboratory Manual For Human Anatomy & Physiology
Biology: Life on Earth
Fundamentals of Anatomy & Physiology Plus Mastering A&P with eText - Access Card Package (10th Edition) (New A&P Titles by Ric Martini and Judi Nath)
Human Anatomy & Physiology
Microbiology with Diseases by Taxonomy (5th Edition)
Human Anatomy & Physiology (11th Edition)
- Figure 7.11 Dinitrophenol (DNP) is an "uncoupler" that makes the inner mitochondrial membrane "leaky" to protons. It was used until 1938 as a weight-loss drug. What effect would you expect DNP to have on the change in pH across the inner mitochondrial membrane? Why do you think this might be an effective weight-loss drug?arrow_forwardcreate a concept map for this. Include all terms. Oxidative Phosphorylation (ETC) Inner Mitochondrial Membrane, Membrane Proteins, High Energy Electrons, NADH, NAD+, FADH, FAD+, ADP, ATP, O2, H2O, ATP Synthase, Proton Gradient Use all terms listed. Make as many connections as you can. Be sure your maps are clear.arrow_forwardPlease explain the Electron Transport System how you understand it. Explanation The electron transport chain is a series of electron transporters embedded in the inner mitochondrial membrane that shuttles electrons from NADH and FADH2 to molecular oxygen. In the process, protons are pumped from the mitochondrial matrix to the inter membrane space, and oxygen is reduced to form water. Question: Respond to the Explanation post above about the electron transport chain.arrow_forward
- The role of a subunit of F0 (2 choices are correct) Undergoes multiple conformational changes from lose to tight to open, to synthesize ATP. Uses two half-channels to pick up and release protons. Allows for unidirectional pick up of protons by c subunit. Allows for the import of inorganic phosphate into mitochondrial matrix.arrow_forwardHi can you please answer to those questions explain this to me , if possible giving examples . Thanks A 45 year old man was rushed to the ER after ingesting "Galaxodin," a hypothetical poison. Explain why Galaxodin is toxic knowing that it increases the pH of the intermembrane space of mitochondria! You will need to give a comprehensive explanation of the process of energy conversion starting from NAH ending in ATP under normal conditions. Include details such as the transfer of electrons, the proton gradient, and chemiosmosis, NADH and FADH2, and ATP synthase must be mentioned. The majority of your grade will come from this part. Then explain why Galaxodin is so toxic!arrow_forwardDraw a simple word diagram showing the path of electrons through the electron-transport chain.b) On your word diagram below, circle the complexes that pump protons across the inner mitochondrial membrane. Put a box around mobile electron carriers.arrow_forward
- The maintenance of a proton motive force across the inner mitochondrial membrane is crucial for continued ATP production. Surprisingly, it has been discovered that the inner membranes of certain cells contain proteins, called uncoupling proteins, that are capable of transporting protons from the intermembrane space to the mitochondrial matrix. Why would mitochondria contain transporters that essentially waste energy potential in the proton gradient?arrow_forwardWhich statement about the ETC is/are FALSE? A. Cytochrome c is freely moving and faces the mitochondrial matrix B. The four respiratory complexes move within the mitochondrial membrane C. CoQ & cytochrome c are not part of the complex but are e- carriers D. CoQ is freely moving within the hydrophilic part of the lipid component of the membranearrow_forwardDraw and label a lipid bilayer containing the large mitochondrial trans-membrane protein complexes representing complex I, II, III, and IV, and ATP Synthase. Label your diagrams with the correct names for the membranes, Make two more of these drawings. Label the first one mitochondrial electron source and using a different ink color, indicate the source(s) of electrons. Label the second mitochondrial energy source, and indicate the source of energy driving the electron transport chain. In the third, label the mitochondrial electron acceptor in its proper position. Also include ATP synthase (in its correct orientation-the spherical part is on the opposite side of the membrane as the H+ reservoir). Use a different color of ink to show where the protons (H+) are pumped to drive ATP synthesis. also indicate where ATP is generated and were the power is stored to generate ATP. NOW, do the same thing with chloroplasts. Draw and label a lipid bilayer containing the large photosynthetic…arrow_forward
- Help is appreciated. Think of an unusual eukaryotic species in which its ATP synthase has 12 subunits for its c ring. How many ATP molecules can be produced per pair of electrons that traverse the respiratory chian starting at Complex I? A. 2 ATP/2e- B. 1 ATP/2e- C. 0.5 ATP/2e- D. 1.5 ATP/2e- E. 3 ATP/2e-arrow_forwardCyanide is a poisonous substance. Cyanide is a cytochrome c oxidase inhibitor that is competitive. The last enzyme of the electron transport chain in mitochondria is cytochrome c oxidase. One of the most common substrates for cytochrome c oxidase is oxygen.Would an increase in the concentration of oxygen affect the inhibitioncaused by cyanide?How would conditions in the mitochondrial intermembrane spacechange if cytochrome c oxidase and the electron transport chain are notfunctioning properly.. Explain why this change in conditions will be harmful to a cell.arrow_forwardThe higher the altitude the lower the oxygen level in air. Climbers of very tall mountains risk altitude sickness which is characterized by shortness of breath, weakness, dizziness, and confusion. The early symptoms of cyanide poisoning are the same as those for altitude sickness. Cyanide binds tightly to cytochrome c oxidase, a protein complex that is the last component of the mitochondrial electron transfer chain. Cytochrome c oxidase with bound cyanide can no longer transfer electrons. Explain why cyanide poisoning starts with the same symptoms as altitude sickness.arrow_forward
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning
Biology: The Dynamic Science (MindTap Course List)BiologyISBN:9781305389892Author:Peter J. Russell, Paul E. Hertz, Beverly McMillanPublisher:Cengage Learning

