
(a)
Interpretation: The transition state of the given reaction is to be interpreted for the given conversion:
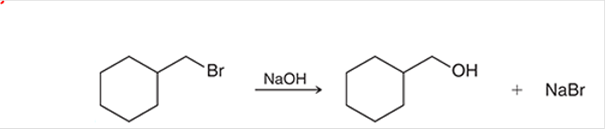
Concept introduction:
(b)
Interpretation: The transition state of the given reaction is to be interpreted for the given conversion:
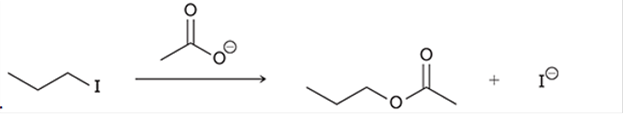
Concept introduction:
Haloalkanes can show the nucleophilic substitution reactions. There are two possible mechanisms
(c)
Interpretation: The transition state of the given reaction is to be interpreted for the given conversion:
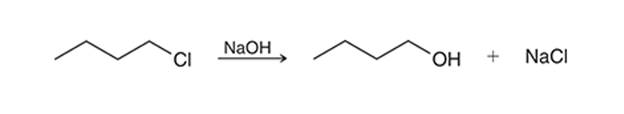
Concept introduction:
Haloalkanes can show the nucleophilic substitution reactions. There are two possible mechanisms
(d)
Interpretation: The transition state of the given reaction is to be interpreted for the given conversion:
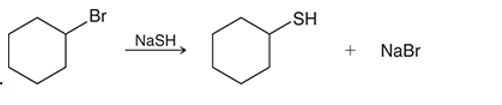
Concept introduction:
Haloalkanes can show the nucleophilic substitution reactions. There are two possible mechanisms
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
ORGANIC CHEMISTRY-STUD.SOLNS.MAN+SG(LL)
- Predict the elimination products for the reaction. Identify the major and minor stereoisomers and draw them as indicated. H₂O Major and Minor isomers -Br H H3C Draw the major isomer. Draw the minor isomer. Select Draw Rings More Select Draw Rings Erase с H CH3 Erase Q2 Q More с H O Q2 Qarrow_forwardWhat is the correct reaction intermediate forthe following reactions?arrow_forwardPropose a plausible mechanism for the following transformation: HO [H3O+] EtOH 19.55 The first three steps of the mechanism involve the formation of The first step is The second step is The third step is eTextbook and Media Save for Later Attemarrow_forward
- Can you please help me draw all isomers of this compound and what is the relationships between these isomers, which isomers would react faster or slower?arrow_forwardWhich of the reactions shown below are correct? & OH oxo H₂O* CO₂H CO₂Et CO₂Et NaOCH₂CH3 HOCH₂CH₂ NaOCH₂CH3 HOCH₂CH3 요 CO₂Et H₂C CH3CO₂Et + CHCO₂Et H₂Carrow_forwardWhich of the following reaction coordinate diagrams represents SN1 and E1 reactions? A B C Darrow_forward
- What are several products for the following reaction with mechanisms included?arrow_forwardDraw the products of each reaction and determine the direction of equilibrium.arrow_forwardDraw ALL the products (including minor products and byproducts) for the following reaction (NO mechanism details needed)arrow_forward
- draw x and yarrow_forwardWhich of the following is not a likely intermediate when the following react? s H 'N H [H3O+] pH 4-5 HO I-ZA H Narrow_forwardAn elevated level of the enzyme alkaline phosphatase (ALP) in human serum is an indication of possible liver or bone disorder. The level of serum ALP is so low that it is very difficult to measure directly. However. ALP catalyzes a number of reactions, and its relative concentration can be determined by measuring the rate of one of these reactions under controlled conditions. One such reaction is the conversion of p-nitrophenyI phosphate (PNPP) to p-nitrophenoxide ion (PNP) and phosphate ion. Control of temperature during the test is very important; the rate of the reaction increases 1.47 times if the temperature changes from 30 C to 37 C. What is the activation energy for the ALP-catalyzed conversion of PNPP to PNP and phosphate?arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax




