
Concept explainers
Interpretation:
To identify each of the following as a oxidation or a reduction.
a.
b.
c.
d.
Concept introduction:
Oxidation occurs when an atom’s oxidation state increases during a reaction. Or loss of electrons.
Reduction occurs when an atom’s oxidation state decrease during a reaction. Or gain of electrons.
Oxidation rules:
1. Oxidation state of an element (in its pure form) is 0.
2. The oxidation state of representatives are their ionic charges.
3. Oxidation state of oxygen is always -2.The peroxide ion is always -1.
4. H with nonmetals (covalent compounds) is given +1oxidation state. Usually in the front of the formula.
5. The sum of the oxidation numbers must be zero for compounds and whatever the charge of the ionic species is.
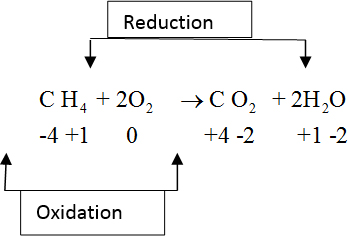
· Oxidation state of “C” in CH4
• Oxidation state of diatomic atoms is zero.
Therefore, O2 oxidation state is zero.
• Oxidation state of “C” in CO2.
• Oxidation state of “O” in H2O.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





