
(a)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.
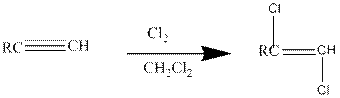
If excess halides are present, a second addition reaction also occurs.
(b)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(c)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(d)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(e)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(f)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry Study Guide and Solutions Manual, Books a la Carte Edition (8th Edition)
- Predict the major product for the following reaction sequence. N-H (two equivalents) ? Modify the given structure of the starting material to draw the major product.arrow_forward6. What is the major product of this reaction? CH3 5 BH3, ,then H₂O₂, OHarrow_forwardWhat is the major product of this reaction?arrow_forward
- Determine the major product of the following reaction OH H3O* H2 Pdarrow_forwardDraw structure of substitution product(s) & major elimination productarrow_forwardWhy won't most primary alkyl halides react in Friedel-Crafts alkylation reactions? O Primary alkyl halides undergo methyl shifts under the standard conditions of Friedel-Crafts alkylation reactions, making them unreactive. Primary alkyl halides create primary carbocations which are too high in energy and too readily undergo rearrangement. Primary alkyl halides do not form a sigma complex after a nucleophilic attack since they connect at the primary carbocation site. Primary alkyl halides as a reactant violates one of the three standard limitations to Friedel-Crafts alkylation reactions.arrow_forward
- I don’t know how to do this questionarrow_forwardFor each of the following reactions predict the major product(s). LOCH3 CI AICI3 HNO3 NH2 1. NaNO2, HCI 2. HBF4, Aarrow_forwardGive the major product of the following reaction. ? 1) NaOH 2) CH;CH,CH,CI, There is no reaction under these conditions or the correct product is not listed here.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
