
BIOLOGY (LL)
5th Edition
ISBN: 9781264115495
Author: BROOKER
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 7.6, Problem 2EQ
CoreSKILL » In the experiment of Figure 7.13, what observation indicated to the researchers that ATP synthase is a rotary machine? What was the control of this experiment? What did it indicate?
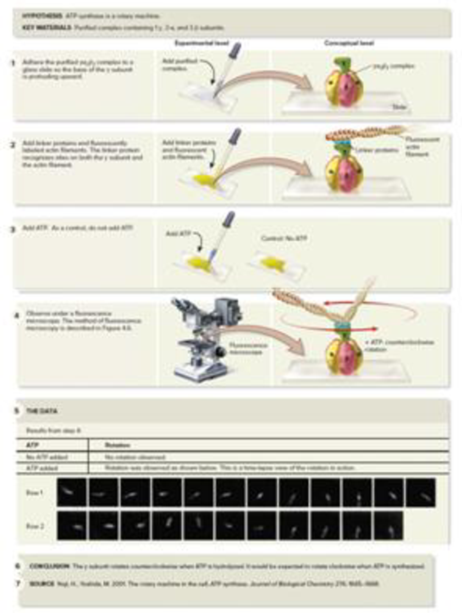
Figure 7.13 Yoshida and Kinosita provide evidence that ATP synthase is a rotary machine.
(5): From Noji, H., Yoshida, M. 2001. The Rotary Machine in the Cell, ATP Synthase. Journal of Biological Chemistry 276: 1665–1668. ©2001 The American Society for Biochemistry and Molecular Biology
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(e) Write 3 assumptions that could be made when using the type of support materials as suggested in (d).
1. a) Describe what is happening in ATP Synthase in plain English! (how would you describe this step for someone who hasn’t taken Biol)
b) If someone wanted to perform an experiment to evaluate the output of a particular step, which variable would be the best option to measure? (Think about which variable is UNIQUE to each step!)
J. C. Servaites, in Plant Physiol. (1985) 78:839–843, observed that Rubisco from tobacco leaves collected before dawn had a much lower specific activity than the enzyme collected at noon. This difference persisted despite extensive dialysis, gel filtration, or heat treatment. However, precipitation of the predawn enzyme by 50% (NH4)2SO4 restored the specific activity to the level of the noon-collected enzyme. Suggest an explanation.
Chapter 7 Solutions
BIOLOGY (LL)
Ch. 7.2 - Core Skill: Connections Look ahead to Table 45.1....Ch. 7.2 - Which organic molecules donate a phosphate group...Ch. 7.2 - Prob. 2CSCh. 7.4 - Prob. 1CCCh. 7.5 - Explain the meaning of the name cytochrome...Ch. 7.6 - Prob. 1CCCh. 7.6 - Prob. 1CSCh. 7.6 - Prob. 1EQCh. 7.6 - CoreSKILL In the experiment of Figure 7.13, what...Ch. 7.6 - Prob. 3EQ
Ch. 7.7 - Prob. 1CCCh. 7 - Which of the following pathways occurs in the...Ch. 7 - Prob. 2TYCh. 7 - Prob. 3TYCh. 7 - Which organic molecule supplies a two-carbon group...Ch. 7 - The ability to diagnose tumors using...Ch. 7 - Prob. 6TYCh. 7 - Certain drugs, which are called ionophores, cause...Ch. 7 - Prob. 8TYCh. 7 - Prob. 9TYCh. 7 - Prob. 10TYCh. 7 - Prob. 1CQCh. 7 - What causes the rotation of the subunit of ATP...Ch. 7 - Core Concept: Energy and Matter How is glucose...Ch. 7 - Discuss the advantages and disadvantages of...Ch. 7 - Prob. 2COQ
Additional Science Textbook Solutions
Find more solutions based on key concepts
Police Captain Jeffers has suffered a myocardial infarction. a. Explain to his (nonmedically oriented) family w...
Human Physiology: An Integrated Approach (8th Edition)
Relative thickness of the myocardium in different chambers; the functional significance of those differences; a...
Anatomy & Physiology: The Unity of Form and Function
Why do scientists think that all forms of life on earth have a common origin?
Genetics: From Genes to Genomes, 5th edition
1. Genetics affects many aspects of our lives. Identify three ways genetics affects your life or the life of a ...
Genetic Analysis: An Integrated Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Practice Mira Gendy 1 of 1 Directions: This short free-response question requires about 6 minutes to answer. The question is worth 3 points. Read the question carefully and completely. Answers must be written out in paragraph form. Outlines, bulleted lists, or diagrams alone are not acceptable. II Substrate Concentration [S] The graph above shows the initial rate of an enzyme-catalyzed reaction at different substrate concentrations in the presence of a constant concentration of the enzyme. Connect the primary structure of the enzyme to its overall shape. I U x X2 5 Initial Rate of Reactionarrow_forwardQ1: Why is it important that enzymes are not permanently altered when they bind with substrate molecules? Q2: How would a higher temperature or higher salt concentration make it more difficult for an enzyme to function effectively? Q3: If a cell was unable to produce a particular enzyme necessary for a metabolic pathway, describe how the absence of that enzyme would affect the cell.arrow_forwardurgenttttttarrow_forward
- what is the relation between K'eq and mass action ratio (Q) for biochem? specifically what are the signs of delta G and delta G' and when is the reaction near equilibriumarrow_forwardGraph B above depicts Lineweaver-Burk double reciprocal plot for an enzyme catalyzed reaction carried out in the presence or absence of an inhibitor. Which of the following statement best describes the kinetic data shown below: ? Line 1 depicts the enzyme-catalyzed reaction carried out in the presence of a competitive inhibitor. Line 1 depicts the enzyme-catalyzed reaction carried out in the presence of a noncompetitive inhibitor. Line 2 depicts the enzyme-catalyzed reaction carried out in the presence of a competitive inhibitor. Line 2 depicts the enzyme-catalyzed reaction carried out in the presence of a noncompetitive inhibitor.arrow_forwardBriefly comment on the differences of using a fixed-time assay versus a kinetic assay to measure enzyme activity. Is it reasonable to assume that the reaction velocity obtained by measuring the amount of product after 30 minutes in a fixed-time assay is directly proportional to absorbance? How could you determine whether this was the case? Word limit 180 words including citation and referencearrow_forward
- Suppose the concentration of glucose inside a cell is 0.1 mM and the cell issuspended in a glucose solution of 0.01 mM.(a) What would be the free energy change, in kJ/mol, for the transport ofglucose from the medium into the cell? Assume T = 37 °C.(b) What would be the free energy change, in kJ/mol, for the transport ofglucose from the medium into the cell if the intracellular and extracellularconcentrations were 1 mM and 10 mM, respectively?(c) If the processes described in parts (a) and (b) were coupled toATP hydrolysis, how many moles of ATP would have to be hydrolysed,per mole of glucose transported, in order to make each process favorable? (Use the standard free energy change for ATP hydrolysis.)arrow_forward.Wheeler and Mathews (J. Biol. Chem. 286:16992–16996, 2011) reported the concentrations of adenine nucleotides in rat liver mitochondria as fol- lows: ATP, 5.5 mM; ADP, 5.1 mM; AMP, 1.8 mM. (a) Calculate the adenylate energy charge within the mitochondrion [ATP] + 0.5[ADP] (adenylate energy charge [ATP] + [ADP] + [AMPJ' (b) Most measurements of adenylate energy charge in whole cells or cytosol give values close to 0.9. Speculate on reasons why it might be advantageous for mitochondria to have an ADP concentration almost as high as that of ATP. (c) succinyl-CoA + ADP + P¡ = -2.9 kJ/mol If [P;] within the mitochondrion is 0.05 M and succinate and succinyl- CoA are present at equimolar concentrations, what is the maximum succinate + ATP + CoA-SH AG°' mitochondrial concentration of CoA-SH at which the reaction can be exergonic?arrow_forwardExercise The following data describe an enzyme-catalyzed reaction. Plot these results using the Lineweaver-Burk method, and determine values for KM and Vinax- The symbol mM represents millimoles per liter; 1 mM = 1 × 10 3 mol L. (The concentration of the enzyme is the same in all experiments.) Velocity (mM sec-") Substrate Concentration (тм) 2.5 0.024 5.0 0.036 10.0 0.053 15.0 0.060 20.0 0.061 Ippt.comarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license