
Introduction to General, Organic and Biochemistry
11th Edition
ISBN: 9781285869759
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 7.7, Problem 7.7P
Problem 7-7
Consider the following equilibrium reaction for the decomposition of an aqueous solution of hydrogen peroxide:
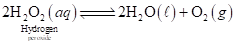
Oxygen has limited solubility in water (see the table in Chemical Connections 6A). What happens to the equilibrium after the solution becomes saturated with oxygen?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
issue 13Balance the reaction below and write the expression for the equilibrium constant.
NO(g) + Cl2(g) ⇌ NOCl(g)I found as a balanced equation: 2NO(g) + Cl2(g) ⇌ 2NOCl(g) and as Kc= 2.5 x 103.Is it correct ?
A reaction occurs according to the equation
2A = Y + 2 Z
If in a volume of 5 dm3 we start with 4 mol of pure A and find that 1 mol of A remains at equilibrium, what is the equilibrium constant Kc?
Answer: [2.7 mol dm-3 ]
K=4.9x10-3 for the reaction CO2(g)+H2(g)⇄ CO(g)+H2O(g). If [CO2]=0.00025 M, [H2]=0.0010 M, & [CO]=0.000015 M, what is the concentration at equilibrium of H2O?
Answer
8.2x10-5M
I got 1.84x10^-14? Can someone explain this to me please.
Chapter 7 Solutions
Introduction to General, Organic and Biochemistry
Ch. 7.1 - Problem 7-1 In the reaction we measure the...Ch. 7.4 - Problem 7-2 Calculate the rate for the reaction in...Ch. 7.6 - Prob. 7.3PCh. 7.6 - Prob. 7.4PCh. 7.6 - Prob. 7.5PCh. 7.7 - Prob. 7.6PCh. 7.7 - Problem 7-7 Consider the following equilibrium...Ch. 7.7 - Prob. 7.8PCh. 7.7 - Prob. 7.9PCh. 7 - 7-10 The rate of disappearance of HCI was measured...
Ch. 7 - 7-11 Consider the following reaction: Suppose we...Ch. 7 - 7-12 Two kinds of gas molecules are reacted at a...Ch. 7 - 7-13 Why are reactions between ions in aqueous...Ch. 7 - Prob. 7.14PCh. 7 - 7-15 A certain reaction is exothermic by 9...Ch. 7 - 7-16 A quart of milk quickly spoils if left at...Ch. 7 - 7-17 If a certain reaction takes 16 h to go to...Ch. 7 - Prob. 7.18PCh. 7 - Prob. 7.19PCh. 7 - Prob. 7.20PCh. 7 - Prob. 7.21PCh. 7 - 7-22 If you add a piece of marble, CaCO3 to a 6 M...Ch. 7 - Prob. 7.23PCh. 7 - Prob. 7.24PCh. 7 - Prob. 7.25PCh. 7 - 7-26 Write the chemical equations corresponding to...Ch. 7 - Prob. 7.27PCh. 7 - 7-28 When the following reaction reached...Ch. 7 - 7-29 The following reaction was allowed to reach...Ch. 7 - Prob. 7.30PCh. 7 - 7-31 Here are equilibrium constants for several...Ch. 7 - 7-32 A particular reaction has an equilibrium...Ch. 7 - Prob. 7.33PCh. 7 - Prob. 7.34PCh. 7 - 7-35 A reaction has a high rate constant but a...Ch. 7 - 7-36 Complete the following table showing the...Ch. 7 - Prob. 7.37PCh. 7 - Prob. 7.38PCh. 7 - Prob. 7.39PCh. 7 - 7-40 Is there any change in conditions that change...Ch. 7 - 7-41 The equilibrium constant at 1127°C for the...Ch. 7 - Prob. 7.42PCh. 7 - 7-43 (Chemical Connections 7A and 7B) Why is a...Ch. 7 - Prob. 7.44PCh. 7 - 7-45 (Chemical Connections 7C) A painkiller—for...Ch. 7 - 7-46 (Chemical Connections 7D) What reaction takes...Ch. 7 - Prob. 7.47PCh. 7 - Prob. 7.48PCh. 7 - Prob. 7.49PCh. 7 - 7-50 Draw an energy diagram for an exothermic...Ch. 7 - Prob. 7.51PCh. 7 - Prob. 7.52PCh. 7 - Prob. 7.53PCh. 7 - Prob. 7.54PCh. 7 - Prob. 7.55PCh. 7 - Prob. 7.56PCh. 7 - 7-57 Write the reaction to which the following...Ch. 7 - Prob. 7.58PCh. 7 - Prob. 7.59PCh. 7 - Prob. 7.60PCh. 7 - Prob. 7.61PCh. 7 - Prob. 7.62PCh. 7 - Prob. 7.63PCh. 7 - 7-64 As we shall see in Chapter 20, there are two...Ch. 7 - Prob. 7.65PCh. 7 - Prob. 7.66PCh. 7 - Prob. 7.67PCh. 7 - Prob. 7.68PCh. 7 - 7-69 Pure carbon exists is several forms, two of...Ch. 7 - Prob. 7.70PCh. 7 - 7-71 You have a beaker that contains solid silver...Ch. 7 - Prob. 7.72PCh. 7 - Prob. 7.73PCh. 7 - Prob. 7.74PCh. 7 - Prob. 7.75PCh. 7 - Prob. 7.76PCh. 7 - Prob. 7.77PCh. 7 - Prob. 7.78PCh. 7 - Prob. 7.79PCh. 7 - Prob. 7.80PCh. 7 - Prob. 7.81PCh. 7 - 7-82 An equilibrium mixture of O2, SO2, and SO3...Ch. 7 - Prob. 7.83PCh. 7 - Prob. 7.84P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A chemical equilibrium A ⇄ 2B has a forward rate constant, kf = 12 M –1 s–1, and a reverse rate constant, kr = 18 s–1. If the system has a concentration of [A] = 0.15 M at equilibrium, what is the concentration of B at equilibrium? (no scientific notation)arrow_forwardFor the reaction 2HI(g) <--> H2(g) + I2(g) Kc = 0.224 at 400K If 0.750 moles of HI are placed in a 5.00L flask and allowed to react , what is the [H2] when equilibrium is reached? (ICE problem. perfect square)arrow_forwardReaction: 1 A + 1B → 3C The initial [A] is 0.80 M and [B] is 4.90 M. At equilibrium, [A] is 0.40 M. What is the value of K? Answer: 19.44 (0.96)arrow_forward
- pls answer d, e, f thank u 2NO(g)+O2(g)⇌2NO2(g) + Heat a) where will the reaction be shifted (right or left) if there is an increase in temperature? b) increase in pressure c) increase in volume d.) addition of catalyst e.) cooling the system f.) What happens to the concentration of NO2 if there is a decrease in pressure? g.) removal of NO2 h.) adding more O2 i.) removal of NO?arrow_forwardThe equilibrium constant for the reaction 2 HF (g) ⇌ H₂ (g) + F₂ (g) is 0.360 at a particular temperature. What is the equilibrium constant for the equation ½ H₂ (g) + ½ F₂ (g) ⇌ HF (g)? ANswer _____arrow_forwardissue 9For the reaction below, indicate whether the equilibrium tends "to the left", "to the right" or "no effect" when the pressure increases. Al3+(aq) + 3 OH-(aq) ⇌ Al(OH)3(s) issue 10For the reaction below, indicate whether the equilibrium tends "to the left", "to the right" or "no effect" when the pressure is increased by adding helium gas. PCl5(g) ⇋ PCl3(g) + Cl2(g) ΔH = 56 kJarrow_forward
- What is the equilibrium constant for the following reaction, if [HBr] = 2.5 M, [H2] = 0.5 M and [Br2] = 0.5 M at equilibrium? H2(g) + Br2(g) -----> 2HBr(g) Group of answer choices 50 0.02 0.04 25arrow_forwardConsider the following reaction in which K = 8.3 x 10-2 at 250˚C.At equilibrium, C = 0.52 mol and [O2] = 0.75 M. What is the equilibrium concentration of CO?2C(s) + O2(g) <---> 2CO(g) Answer is 0.25 M, just want to see steps!arrow_forwardSolid Ca3(PO4)2 is placed into 10.0 L of water. When equilibrium is established, the concentration of Ca²+ is 2.3 x 10-4 M. What is Kc for this equilibrium? Ca3(PO4)2 (s) 3 Ca²+ (aq) + 2 PO4³- (aq) A) 7.7 Correct. 10-5 B) 3.5 × 10-8 C) 2.7 x 10-19 D) 7.2 x 10-20arrow_forward
- For the following reaction 3O₂(g) ⇌ 2O₃(g) Kc = 2.10 × 10⁻⁷ at a certain temperature. If [O₂] = 0.0365 M when at equilibrium, what is the equilibrium O₃ concentration? Answer ___ Marrow_forwardCalculate the equilibrium constant of the reaction CO(g) + 2 H2S(g) CS2(g) + H2O(g) + H2(g) from the following information: CO(g) + 3 H2(g) CH4(g) + H2O(g) K = 9.17 x 10-2 CH4(g) + 2 H2S(g) CS2(g) + H2(g) K = 3.3 x 104 Answer: [3.03 x 103 ]arrow_forwardAt 2000 K, the equilibrium constant, Kc for the decomposition of hydrochloric acid is 3.2 x 10-3. If the initial concentration of HCl is 0.250 M, what are the equilibrium concentrations of hydrochloric acid, hydrogen gas, and chlorine gas? Show all work. 2HCl(g) ⇄ H2(g) + Cl2(g)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY