
Interpretation:
The name and stability (most stable to least stable) of given
Concept Introduction:
Delocalized electrons: Electrons that are not localized in one particular atom or bond and shared by three or more atoms are called delocalized electrons.
Compounds with two double bonds are known as dienes.
Stability of diene: The number of alkyl group increases with increasing the stability of the dienes.
Conjugated diene: Dienes are hydrocarbons with two double bonds; where the conjugated double bonds are separated by one single bond.
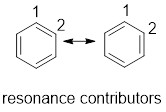
Resonance contributor: In some chemical compounds like benzene pi electrons are delocalized in it and difficult to locate it. Resonance contributor gives an idea of whereabouts of pi electrons. The exact structure with localized electrons are called resonance contributor.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Essential Organic Chemistry, Global Edition
- When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.Show why A predominates at -15 °C and B predominates at 60 °C.arrow_forwardIn 4+2 cycloaddition, ENDO stereochemistry is preferred because: endo stereochemistry allows the diene to adopt s-cis conformation. endo stereochemistry results in less steric hindrance. the transition state leading to endo stereochemistry is higher energy. it allows for additional π to π overlap between the diene and the dienophile.arrow_forwardIs the following alkene E,Z, or neither? a. E b. Z c. neither d. It is one or the other, but which is impossible to determine from the information givenarrow_forward
- The central carbon atom of an allene is a member of two double bonds, and ithas an interesting orbital arrangement that holds the two ends of the molecule at rightangles to each other.(a) Draw an orbital diagram of allene, showing why the two ends are perpendicular.(b) Draw the two enantiomers of penta-2,3-diene. A model may be helpfularrow_forwarda. Show that [4+4] cycloaddition of two butadiene molecules to give cycloocta-1,5-diene is thermally forbidden but photochemically allowed b.Thermally allowed cycloaddition of the two butadiene molecules when there's a different. Show reaction and explain why it is thermally allowarrow_forwardExplain the Carbocation Rearrangements ?arrow_forward
- This is a Diels-Alder reaction between ethylene and cis-1,3-butadiene. For each cycloaddition product, draw in all hydrogen atoms, and write the molecular formula below.arrow_forwardHow many linear dienes have molecular formula C6H10? (Disregard cis–trans isomers.) A. How many of the linear dienes in part a are conjugated dienes? B. How many are isolated dienes? C. How many are cumulated dienes?arrow_forwardWhat is the best way to draw a molecular orbital diagram for 1,3-cyclopentadiene, identifying the Lowest unoccupied molecular orbital and the highest occupied molecular orbital?arrow_forward
- Draw an approximate energy diagram withannotations for the rotamers of 1,2-dibromoethanefrom 0-180°. Give the Newman projections andnames of the most important rotamers.arrow_forwardRank the following dienes from most reactive to least reactive in a Diels–Alder reaction:arrow_forward1,3-pentadiene reacts with HBr. When the double bond at the 3-position reacts, the hydrogen attaches to the 4-carbon, thus forming an allylic carbocation with delocalized electrons. This carbocation has another resonance structure. Because there are equal amounts of diene and hydrogen bromide, in which molecule only one of the double bonds reacts. Which of the following correctly describes this new resonance structure? a) It has a double bond between carbons 3 and 4 b) It has a positive charge on carbon 2 c) It has a positive charge on carbon 4 d) It has a double bond between carbons 2 and 3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





