
Concept explainers
Interpretation:
The mechanism for each step in the pathway given for bio synthesizing epi-Aristolochene is to be shown using curved arrows. Further the steps that involve electrophilic addition(s) to the
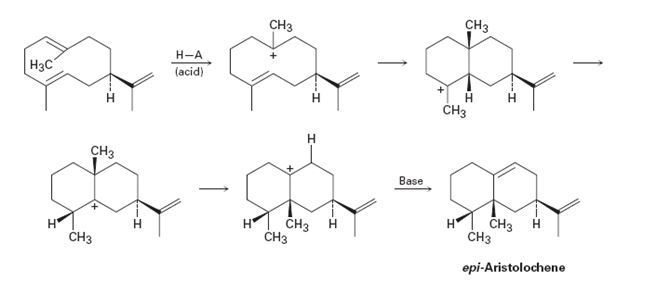
Concept introduction:
While drawing curved arrows the head of the curved arrow starts from the nucleophilic source (either negatively charged or neutral) and ends in an electrophilic sink (either positively charged or neutral). The formation of a new bond should lead to the breaking of a bond that already exists as the octet rule cannot be violated.
In electrophilic addition reactions, the first step is the attack of the π electrons of the double bond on the hydrogen of another reactant to yield a carbocation. One of the carbon in C=C gets attached to hydrogen while the other acquires a positive charge. In the second step, the carbocation formed can rearrange to give another more stable carbocation either by a hydride shift (shift of hydrogen atom with its electron pair) or by an alkyl shift (shift of an alkyl group with its electron pair) between neighboring carbons. In the last step the carbocation produced reacts with a negatively charged ion or eliminates a proton to give the product.
To show:
The mechanism for each step in the pathway given for bio synthesizing epi-aristolochene using curved arrows. Further to identify the steps that involve electrophilicaddition(s) to the alkene and that involve carbocation rearrangement(s).
Trending nowThis is a popular solution!

Chapter 7 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- What is the mechanism and product for the following reaction?arrow_forwardDraw the products of the SN1 reaction below and use wedge-and-dash bonds to indicate the stereochemistry of any stereogenic centers.arrow_forwardDraw the product of an SN2 reaction shown below. Use wedge and dash bonds to indicate stereochemistry where appropriate. Indicate inorganic byproducts.arrow_forward
- A8 Is there anyone who can provide this pericyclic rxn mechanism?arrow_forwardWrite the complete and detailed mechanism for THREE EXAMPLES of SN1 reactions. In each example , clearly name and identify the MOLECULES and PRODUCTS at each stage and use curved arrows to show electron movement at each stage of the reaction.arrow_forwardWhat is the mechanism for the following reaction?arrow_forward
- What are the products obtained (2 stereoisomers A and B) when (Z)-hex-3-ene is treated with Br2 in anhydrous medium? Show the mechanism of this reaction. When the dibromide formed is treated with KOH in EtOH, only (Z)-3-bromohex-3-ene. Explain this result and justify why the E isomer cannot be obtained here. here.arrow_forwardThe bicyclo [3.1.0] hexane ring system, highlighted in compound 3, is found in several natural products, including sabinene, a compound partially responsible for the flavor of ground black pepper. One method for preparing this ring system involves the conversion of compound 1 to compound 2, as shown below. Draw the structure of compound 2 and provide a reasonable mechanism for its formation. Add any remaining curved arrows to complete step one of the mechanism, and modify the given drawing as needed to show the intermediate that is formed in this step.arrow_forwardGive the following reaction A+3B --->2C.arrow_forward
- Draw the products of the following reaction and show the mechanism; CH3-CH2-CCH3Br-CH3 + CH3OHarrow_forwardwhat is the expected major product of HBr addition to the alkene shown below? Show the mechanismarrow_forwardunder what type of mechanism are the following reactions carried out: SN2, SN1, E2, E1? show both reactionsarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


