
Concept explainers
How many
How many yield 2,3-dimethylbutane?
How many yield methylcyclobutane?
Interpretation:
The number of alkenes which are required to produce the given alkanes on catalytic hydrogenation is to be determined.
Concept introduction:
On catalytic hydrogenation, alkenes get converted to the corresponding alkanes with the same number of carbon atoms.
In hydrogenation reaction, one hydrogen atom gets attached to each of the double bonded carbon atoms.
To accelerate the rate of hydrogenation, the metal catalyst provides an alternative pathway involving low activation energy steps.
The alkane that is formed contains two hydrogen atoms more than the corresponding alkene and these two hydrogen atoms are added on the adjacent carbon atoms in the alkene.
Answer to Problem 25P
Solution:
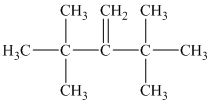
a) Only one alkene can produce
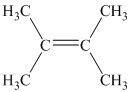
b) Two alkenes can produce
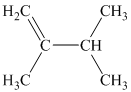
c) Three alkenes can produce methylcyclobutane upon catalytic hydrogenation.
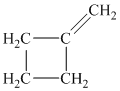
Explanation of Solution
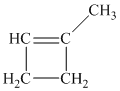
a) The structure of
Hydrogenation of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to at least one hydrogen atom each.
In
The alkane structure is symmetric, and carbon atoms
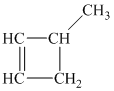
b) The structure of
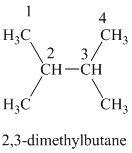
The hydrogen of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to at least one hydrogen atom each.
In
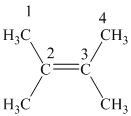
The
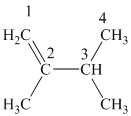
The alkane structure is symmetric, and the carbon atoms
Hence, two alkenes can produce
c) The structure of methylcyclobutane is shown below:
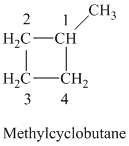
The hydrogen of alkenes adds one hydrogen atom on each carbon atom of the double bonded carbon atoms. So in the alkane, these two carbon atoms (previously double bonded) should be attached to one at least one hydrogen atom each.
In methylcyclobutane, the
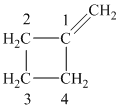
The
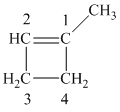
The
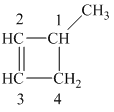
The alkane structure is symmetric. This means, there cannot be any other distinct alkene structure which would produce methylcyclobutane upon catalytic hydrogenation.
Hence, three alkenes can produce methylcyclobutane upon catalytic hydrogenation.
Want to see more full solutions like this?
Chapter 8 Solutions
ORGANIC CHEMISTRY(LL)-W/ACCESS >CUSTOM<
- (1R,2R)-1-Bromo-2-methylcyclopentane is reacted with sodium methoxide. Given the product(s) and show the reaction mechanism, including the depiction of the transition state. Draw an energy diagram for this conversion.arrow_forwardWrite down all possible alkene products from the following Elemination reaction (no mechanism is required) indicate which one will be major product. And explain it?arrow_forward1/ For each of the molecular formulas, determine the number of elements of unsaturation and draw 3 examples: a) C6H8O2 b) C6H3NClBr 2/ Propose mechanism (showing explicity how observed mixtures of products are formed) cyclopenta-1,3-diene + Br2 ---> 3,4-dibromocyclopent-1-ene + 3,5-dibromocyclopent-1-ene 3/ Diel-Alder product of heating hexa-1,3,5-triene with maleic anhyarrow_forward
- Rank the following alkenes in order of stability.arrow_forwardDraw mechanism arrows and then predict the major products and stereochemistry. Also include the labels of thermodynamic and kinetic no stick structure don't understand 3,4-dimethylhex-3-ene with BH3. After reaction is complete, add H2O2, –OH, and H2Oarrow_forwardThe following Grignard reagents are desired: a) cyclohexylmagnesium bromide b) sec-butylmagnesium bromide but only alkenes are available as raw material. Write the reaction equations that lead to the desired products, including the reaction conditions. Remember that if it is about discrete steps you must make it clearly noticeable.arrow_forward
- Show the structures for the major/significant organic product(s) expected for the following eleven reactions or sequences of reactions. If there is a major and minor productcircle the major product. Show all relevant stereochemistry.arrow_forwardShow stepwise mechanism by pushing arrows (fishhook) and drawing ALL intermediates. Draw a detailed mechanism for the addition of HBr to 3,3-dimethyl-1-butene in the presence of dimethlyperoxide. Show and label all steps including, initiation, propagation, and termination.arrow_forwardDraw mechanism arrows and then predict the major products and stereochemistry. Also include the labels of thermodynamic and kinetic 3,4-dimethylhex-3-ene with HClarrow_forward
- Determine the predominant type of reaction: SN1, SN2, E1 and E2. Draw the major products only. Show stereochemistry whenever applicable.arrow_forwardThe substitution reaction below forms two ethers (which are consitutional isomers). Which ether is the major product and why?arrow_forwardRank the following alkenes in order of increasing stability (least to most stable)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
