
Concept explainers
Interpretation:
The Lewis structures of the given molecules and ions are to be represented.
Concept Introduction:
In Lewis dot
In Lewis dot symbol, valence electrons are represented by dots.
Dots are placed above and below as well as to the left and right of symbol.
Number of dots is important in Lewis dot symbol but not the order in which the dots are placed around the symbol.
In writing symbol pairing is not done until absolutely necessary.
For metals, the number of dots represents the number of electrons that are lost when the atom forms a cation.
For second period non metals, the number of unpaired dots is the number of bonds the atom can form.
Atomic ions can also be represented by dot symbols, by simply adding (for anions) and subtracting (for cations) the appropriate number of dots from Lewis dot symbol.
Lewis structure is the representation of bonding and non-bonding electron pairs present in the outermost shell of all atoms present in the molecule.
The number of bonds formed by an atom in the molecule is determined by the valence electron pairs.
Answer to Problem 42QP
Solution:
(a)
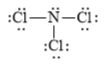
(b)
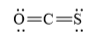
(c)
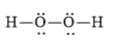
(d)
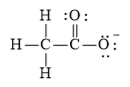
(e)
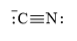
(f)
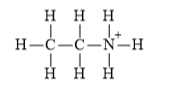
Explanation of Solution
a)
The electronic configuration of nitrogen and chlorine in
The nitrogen atom contains three valence electrons in its
The Lewis structure of
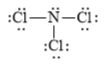
b)
The electronic configuration of oxygen, carbon, and sulfur in
Oxygen and sulfur atoms contain two valence electrons in their
The Lewis structure of
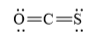
c)
The electronic configuration of oxygen and hydrogen in
The oxygen atom contains four valence electrons in its
The Lewis structure of
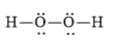
d)
The electronic configuration of oxygen, carbon, and hydrogen in
The carbon atom has a tendency to form four bonds because of the presence of four valence electrons in its outermost shell, hydrogen has a tendency to form one bond because of the presence of one electron in its outermost shell, and oxygen has a tendency to form two bonds due to the presence of two electrons in its outermost shell.
The Lewis structure of

e)
The electronic configuration of nitrogen and carbon in
Cyanide ion is composed of one triple bond of carbon and nitrogen atom. This species contains one lone pair on both carbon and nitrogen atoms.
The Lewis structure of
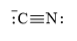
f)
The electronic configuration of carbon, nitrogen, and hydrogen in
Carbon atom has a tendency to form four bonds because of the presence of four valence electrons in its outermost shell, hydrogen has a tendency to form one bond because of the presence of one electron in its outermost shell, and nitrogen has tendency to form four bonds due to the presence of three electrons in its
The Lewis structure of

Want to see more full solutions like this?
Chapter 8 Solutions
Combo: Chemistry With Connect Access Card With Learnsmart And Learnsmart Labs Access Card
- Acetyl chloride, CH₃C(O)Cl, is used as a reagent for the acylation of salicylic acid in the synthesis of aspirin. Draw the Lewis structure of CH₃C(O)Cl (with minimized formal charges) and then determine if the molecule is polar or nonpolar. +arrow_forwardWhat's the written lewis structure for C2H5O2N and how many valence electrons?arrow_forwardUsing the bond dissociation enthalpies in Table 8.8, estimate the enthalpy of combustion of gaseous methane, CH4, to give water vapor and carbon dioxide gas.arrow_forward
- Write all resonance structures of chlorobenzene, C6H5Cl, a molecule with the same cyclic structure as benzene. In all structures, keep the CCl bond as a single bond. Which resonance structures are the most important?arrow_forwardAn ionic compound has the following composition (by mass): Ca, 30.3%; N, 21.2%; O, 48.5%. What are the formula and name of the compound? Write the Lewis formulas for the ions.arrow_forwardA liquid compound used in dry cleaning contains 14.5% C and 85.5% Cl by mass and has a molecular mass of 166 amu. Write the Lewis formula for the molecule.arrow_forward
- Nitrosyl azide, N4O, is a pale yellow solid first synthesized in 1993. Write the Lewis structure for nitrosyl azide.arrow_forwardA gaseous compound has the following composition by mass: C, 25.0%; H, 2.1%; F, 39.6%; 0. 33.3%. Its molecular mass is 96.0 amu. Write the Lewis formula for the molecule.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



