
(a)
Interpretation:
The more reactive compound should be identified from the given pair compounds for an E1 reaction.
Concept introduction:
E1 reaction is unimolecular reaction. The rate of the reaction is depends upon the nature of
Stability of carbocation is in the order of,
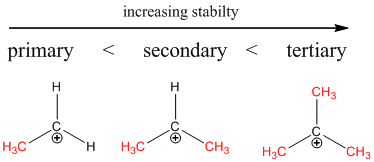
The stability order is on the basis of hyperconjugation. Hypercongugation is the process of delocalization of electrons through parallel overlap of p-orbital and hybridized orbital.
The more number of alkyl attachment on carbocation makes more hyperconjugation in that system. Hence the tertiary carbocation will show more hyperconjugation.
To identify: the more reactive compound in E1 reaction from the pair of given compounds.
(b)
Interpretation:
The more reactive compound should be identified from the given pair compounds for an E1 reaction.
Concept introduction:
E1 reaction is unimolecular reaction. The rate of the reaction is depends upon the nature of alkyl halide. Carbocation is formed as an intermediate. Stability of carbocation determines the rate of reaction.
Stability of carbocation is in the order of,

The stability order is on the basis of hyperconjugation. Hypercongugation is the process of delocalization of electrons through parallel overlap of p-orbital and hybridized orbital.
The more number of alkyl attachment on carbocation makes more hyperconjugation in that system. Hence the tertiary carbocation will show more hyperconjugation.
To identify: the more reactive compound in E1 reaction from the pair of given compounds.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





