
Concept explainers
Introduction:
Organisms are made up of carbon based molecules. Life on Earth is often considered carbon- based.
Answer to Problem 6STP
Correct answer :
The correct answer is option (C), C6H12O4.
Explanation of Solution
Explanation/justification for the correct answer:
Option (C). C6H12O4− To write the molecular formula of a compound, first we have to count the number of individual atoms of each element. These numbers are written as subscripts of each atom’s symbol. Structure of the given compound is as follows.
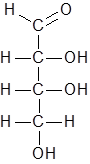
Number of carbon atoms= 4
Number of hydrogen atoms= 8
Number of oxygen atoms= 4
If we add two more carbon atoms, number of carbon atoms= 6
To satisfy the valency of carbon, each carbon atom will combine with 2 more hydrogen atoms. So there will be 12 hydrogen atoms. Number of oxygen atoms will remain same as 4.
Hence the new structure is as follows:
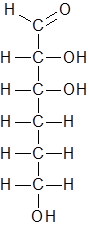
The molecular formula of this compound is C6H12O4
Explanation for incorrect answer:
Option (A). C6H8O4− Structure of the given compound is as follows.
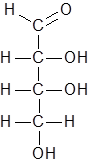
Number of carbon atoms= 4
Number of hydrogen atoms= 8
Number of oxygen atoms= 4
If we add two more carbon atoms, number of carbon atoms= 6
To satisfy the valency of carbon, each carbon atom will combine with 2 more hydrogen atoms. So there will be 12 hydrogen atoms. Number of oxygen atoms will remain same as 4.
Hence the new structure is as follows:
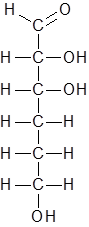
The molecular formula of this compound is C6H12O4 Hence this is not the correct option.
Option (B). C6H10O6- Structure of the given compound is as follows.
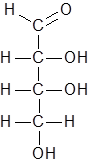
Number of carbon atoms= 4
Number of hydrogen atoms= 8
Number of oxygen atoms= 4
If we add two more carbon atoms, number of carbon atoms= 6
To satisfy the valency of carbon, each carbon atom will combine with 2 more hydrogen atoms. So there will be 12 hydrogen atoms. Number of oxygen atoms will remain same as 4.
Hence the new structure is as follows:
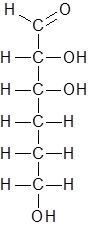
The molecular formula of this compound is C6H12O4 Hence this is not the correct option.
Option (D).C6H12O6- Structure of the given compound is as follows.
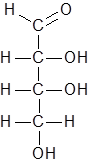
Number of carbon atoms= 4
Number of hydrogen atoms= 8
Number of oxygen atoms= 4
If we add two more carbon atoms, number of carbon atoms= 6
To satisfy the valency of carbon, each carbon atom will combine with 2 more hydrogen atoms. So there will be 12 hydrogen atoms. Number of oxygen atoms will remain same as 4.
Hence the new structure is as follows:
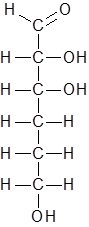
The molecular formula of this compound is C6H12O4.
Hence this is not the correct option.
Chapter 8 Solutions
Biology Illinois Edition (Glencoe Science)
Additional Science Textbook Solutions
Microbiology with Diseases by Body System (4th Edition)
Anatomy & Physiology (6th Edition)
Campbell Biology: Concepts & Connections (9th Edition)
Fundamentals of Anatomy & Physiology (11th Edition)
Human Physiology: An Integrated Approach (7th Edition)
Campbell Biology (11th Edition)
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





