
Concept explainers
(a)
Interpretation:
Using curved arrow notation, an
Concept introduction:
The
Answer to Problem 8.1P
Mechanism for the given
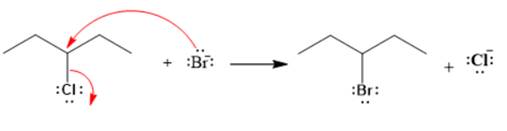
Explanation of Solution
The given reaction is
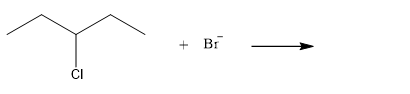
The given substrate is a secondary alkyl halide. Chlorine is a moderately good leaving group. Bromide ion is a strong nucleophile. An 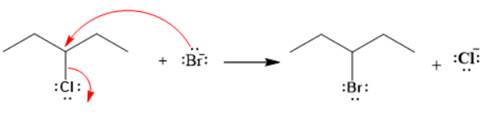
In the above reaction, the bromide ion attacks the carbon, to which the leaving group, chlorine, is attached from the side opposite the leaving group. In the product, the chlorine atom is replaced by a bromine atom.
The
(b)
Interpretation:
Using curved arrow notation, an
Concept introduction:
The
Answer to Problem 8.1P
Mechanism for the given
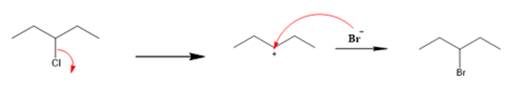
Explanation of Solution
The given reaction is
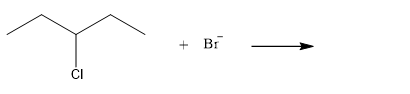
The first step is the breaking of carbon-chlorine bond. This will generate a secondary carbocation intermediate. In the second step, the electron rich
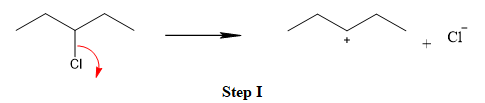
The leaving group is lost and forms a carbocation as follows:
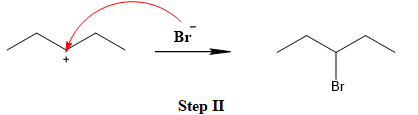
The
Want to see more full solutions like this?
Chapter 8 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- Please draw a curved arrow mechanism for the following reaction. You may use generic acids and bases to protonate and deprotonate when appropriate, but it is in ACIDIC solution. m= & Br₂ H+arrow_forwardDraw a curved arrow mechanism for the reaction. You can assume that all reactants and products are shown.arrow_forwardPlease answer this mechanism problem. Please draw out the solutions with arrows and formal charges.arrow_forward
- Draw the simplest curved arrow mechanism possible for the reaction shown below. You may need to re-draw structures to show bond lines or lone pairs.arrow_forward. Use to curved arrow notation, propose a mechanism for the following reaction and state whether it is either SN1, E1, SN2, or E2. Give the IUPAC names of all organic reactants and products.arrow_forwardDraw a mechanism for the reaction of methylamine with formic acid. In the box to the left, draw any necessary curved arrows. Show the products of the reaction in the box to the right. Include any nonzero formal charges and all lone pairs of electrons. Finally, check the box to indicate which side of the reaction is favored at equilibrium.arrow_forward
- Draw the curved arrow mechanism to explain the product formation for the following reactions, show how the reaction is catalytic and name all components.arrow_forwardCan you please answer the question and show the mechanism.arrow_forwardUse curved arrow formalism to show the mechanism of the reaction shown below.arrow_forward
- 2. Draw the complete, detailed El mechanism for the following reaction (including including curved arrows). CH;OH -Brarrow_forwardClassify each reaction as either a proton transfer reaction, or a reactionof a nucleophile with an electrophile. Use curved arrows to show how theelectron pairs move. (file attached)arrow_forwardMechanism. Draw complete arrow-pushing mechanisms for the following reactions.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





