
(a)
Interpretation:
The mechanism for the given elimination reaction including carbocation rearrangement is to be drawn.
Concept introduction:
The
Answer to Problem 8.65P
The
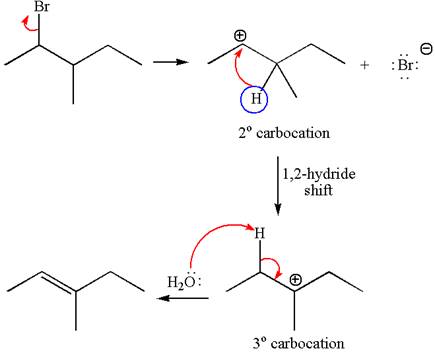
Explanation of Solution
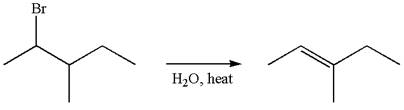
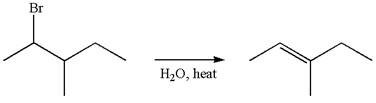
The given reaction equation is
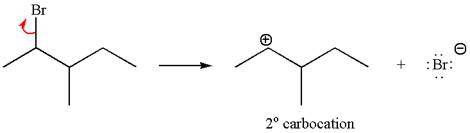
In first step, the leaving group
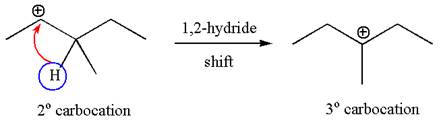
The carbocation formed is rearranged by
The water molecule acts as a base and abstracts a proton from the carbon adjacent to the carbocation, forming
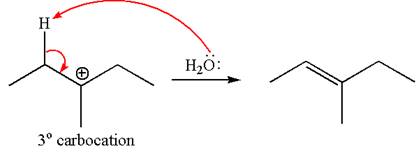
The mechanism for the given elimination reaction is drawn to show the carbocation rearrangement by
(b)
Interpretation:
The mechanism for the given elimination reaction without carbocation rearrangement is to be drawn.
Concept introduction:
The
Answer to Problem 8.65P
The
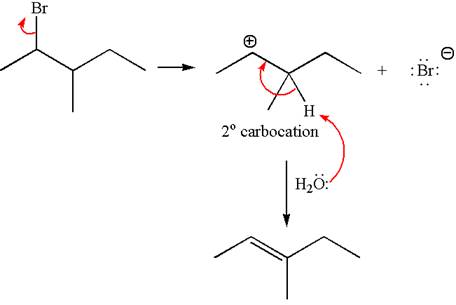
Explanation of Solution
The given reaction equation is
In the first step, the leaving group
In the second step, without rearrangement, the proton is eliminated by the base
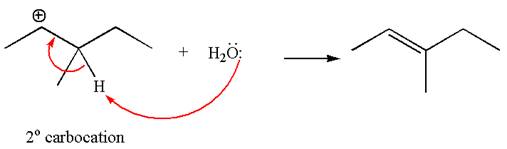
The mechanism for the given elimination reaction is drawn to without rearrangement step, indicating that the same product is formed with or without rearrangement.
(c)
Interpretation:
It is to be explained how the
Concept introduction:
The
Answer to Problem 8.65P
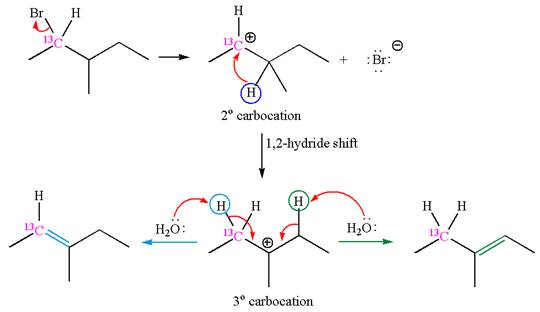
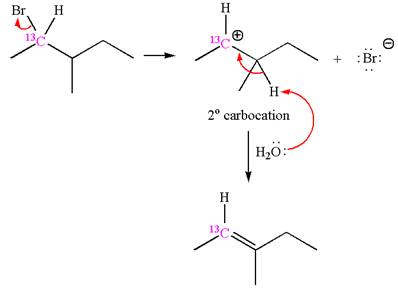
The reaction with carbocation rearrangement gave two products, and the reaction without carbocation rearrangement gave only one product, as shown below, indicating that the
Reaction with rearrangement:
Reaction without rearrangement:
Explanation of Solution
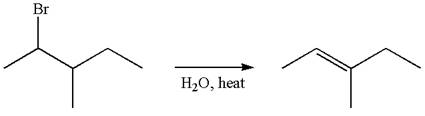
The given reaction equation is
If the carbon bonded to the leaving group in the given substrate is labelled as
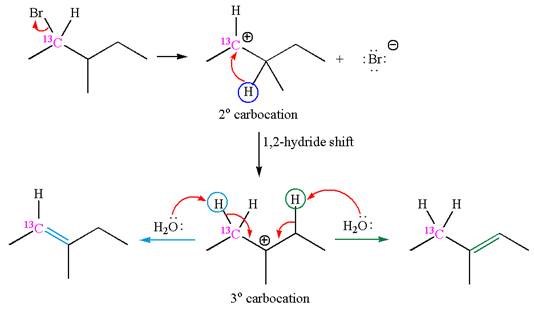
In one product, one of the double bonded carbon is
If the reaction proceeds without rearrangement, then only one product is formed where one of the double bonded carbon is
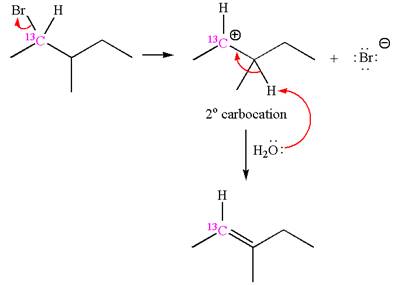
As the reaction with rearrangement of carbocation formed two products, and reaction without rearrangement formed only one product, it indicates that the E1 products depend on whether the rearrangement occurred.
It is explained that the E1 products depend on whether the reaction includes carbocation rearrangement occurring with
(d)
Interpretation:
How the deuterium isotope labeling is useful to determine whether the rearrangement is occurred in given
Concept introduction:
The
Answer to Problem 8.65P
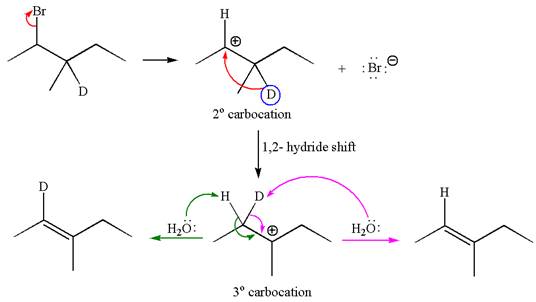
The reaction with carbocation rearrangement by migration of deuterium gave two products, and the reaction without carbocation rearrangement gave only one product as shown below, indicating that the deuterium isotope labeling is useful to determine whether the rearrangement occurred in the given
Reaction with rearrangement:
Reaction without rearrangement:
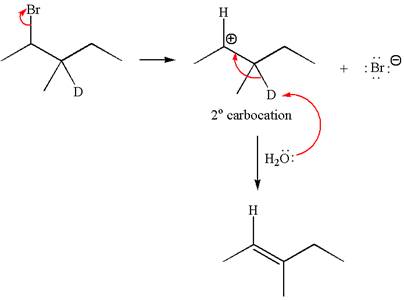
Explanation of Solution
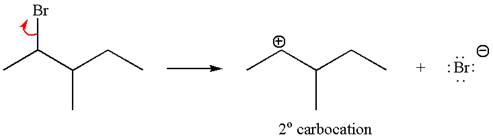
The given reaction equation is
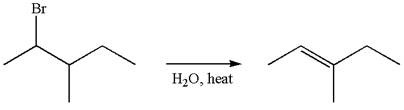
If the migrating hydrogen in the given substrate is replaced with deuterium, then two products are formed when the reaction occurred through carbocation rearrangement. The detailed mechanism is as follows:
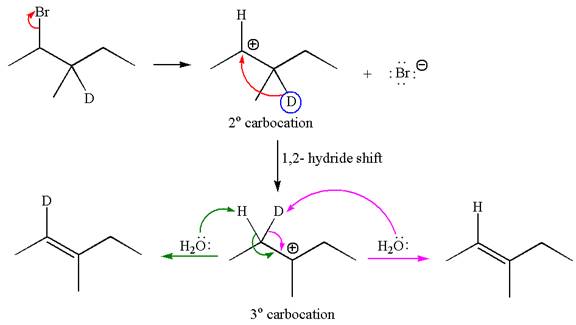
One product is formed by elimination of hydrogen atom and another by elimination of deuterium atom.
If the reaction proceeds without rearrangement, only one product is formed by elimination of deuterium atom. The detailed mechanism is as follows:
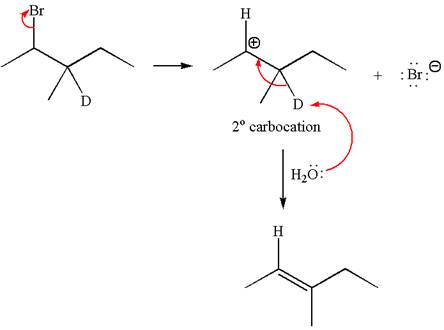
As the reaction with rearrangement of carbocation formed two products and reaction without rearrangement formed only one product, it indicates the E1 products depend on whether the rearrangement occurs.
It is explained on the basis of formation of different products that deuterium isotope labeling is useful to determine whether the rearrangement occurred in the given
Want to see more full solutions like this?
Chapter 8 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- 5. Each of the following may participate in an elimination reaction, under the proper conditions. (a) Circle the alpha (a) carbon. (b) Circle the beta (B) carbon(s). (c) Draw the alkene product(s) that may form, with the new double bond between the a and B positions. (d) If more than one alkene product is possible, circle the most stable (Zaitsev) product. X tx + 3arrow_forwardIn this chapter, reversibility was discussed only in terms of SN2, SN1, E2, and E1 reactions, but the ideas apply to other reactions as well. Considering charge stability, determine whether each of the following elementary steps is reversible or irreversible. (a) (b) (c) H CH3 :? HÖ:arrow_forward5a) Draw the product(s) of the reaction shown below. Additionally, identify the nucleophile, electrophile and leaving group.arrow_forward
- 10) Consider the following alkene hydration reaction. он H2SO, H20 Given that the reaction is endergonic by 10 kcal/mol, complete the following. (a) Draw the intermediate(s) formed in the mechanistic pathway to the major product in the indicated space below. If there are multiple intermediates label them 1, 2, 3, 4, etc. (b) Sketch a reaction-energy profile for this reaction and clearly label staring material, product, intermediate(s), transition state(s), AG" and AG (of the rate determining step) on your plot. Progress of the reaction Draw and Label Intermediate(s) Below: Potential energyarrow_forward6) Write the mechanism (two propagation steps only, starting with the bromine radical in the first step and using Br2 in the second step) and that would explain how the following two products are produced. (Hint – the allylic radical is resonance stabilized.) Br Br NBSarrow_forwardIdentify the carbocations that are likely to rearrange, and state whether rearrangement will proceed via 1,2-methyl shift, or a 1,2-hydride shiftarrow_forward
- Draw the reactants and complete the mechanism by drawing in the appropriate electron-flow arrows (including stereochemistry) for the following Sy2 reaction of an alkyl bromide. Represent the nucleophile as an ion. Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the grid and connecting them with bonds, including charges where needed. Indicate the mechanism by drawing the electron-flow arrows on the molecules. Arrows should start on an atom or a bond and should end on an atom, bond, or where a new bond should be created. • View Available Hint(s) + CH N H. S CH H,C C-ÖH CI HC Br HC F [1] Aarrow_forwardConsider the addition of HBr shown here. (a) Draw all four carbocation intermediates possible from protonation of the diene and identify the most stable one. + HBr (b) Draw both halogenated products formed by attack of Br on that carbocation. (c) Which of those products would you expect to be formed in the greatest amount at low temperatures? (d) Which would you expect to be formed in the greatest amount at high temperatures?arrow_forwardWrite the mechanism of the following bromination reaction and explain the regioselectivity of the bromination reaction. Draw the mechanism, transition state, energy diagram, etc. The same reaction with chlorine is not much regioselective in Q1. Explain the decreased regioselectivity of the following chlorination reaction by comparing the bromination reaction in Q1. Draw the mechanism, transition state, energy diagram, etc.arrow_forward
- When treated with NaOH, the bromide below gives an alkene by the E2 mechanism, by elimination of the H atom indicated by the arrow: (a) Draw the Newman projection from which elimination takes place. (b) Draw the mechanism. (c) Draw the product with the proper stereochemistry. (d) Assign the proper stereochemical descriptor to the product. (e) Give the rate equationarrow_forwardFor each reaction, draw the complete mechanism and the major organic product. (Recall that each of these reactions is expected to have a low yield.) (а) HC=CH H,SO4 H20 (b) (c) D3PO4 ? ? H,SO4 → ? H20 HC=CH D20arrow_forwardConsider the attached E2 reaction What happens to the reaction rate with each of the following changes?[1] The solvent is changed to DMF. [2] The concentration of −OC(CH3)3 is decreased. [3] The base is changed to −OH. [4] The halide is changed to CH3CH2CH2CH2CH(Br)CH3. [5] The leaving group is changed to I−.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





