
Interpretation:
How the rate of the reaction between an
Concept Introduction:
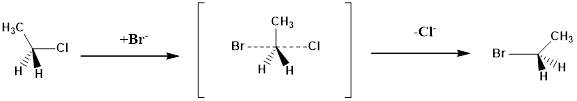
Structure of the substrate plays a major role in
Solvent: it is substance which dissolves the chemical substrate. They are classified as polar protic and
Polar parotic solvent: It contains at least one
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Essential Organic Chemistry (3rd Edition)
- Consider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and (a) Which of the four rings is most reactive in electrophilic aromatic substitution? (b) Which of the four rings is least reactive in electrophilic aromatic substitution? (c) What are the major produces) formed when this compound is treated with one equivalent of Br2?arrow_forwardWhat happens to the rate of the following SN2 reaction when the 3-iodopentane is doubled and the NaCN is tripled?arrow_forwarda. Explain why 1-bromo-2,2-dimethylpropane has difficulty undergoing both SN2 and SN1 reactions. b. Can it undergo E2 and E1 reactions?arrow_forward
- p-Fluoronitrobenzene is more reactive toward hydroxide ion than is p-chloronitrobenzene. What does this tell you about the rate-determining step for nucleophilic aromatic substitution?arrow_forwardWhat direction does the nucleophile approach the substrate in an SN2 reaction?arrow_forwardExplain the observed rate of reactivity of the following 2° alkyl halides in an SN1 reaction.arrow_forward
- How does solvent affect rate of reaction if its increase or decrease in sn1 or sn2arrow_forward1. Put these three common types of carbonyl compound in order of decreasing reactivity ester amide acid chloride 2. For the least reactive, show the interconversion to its other resonance form: How does this electron delocalisation make it stable? 3. For the most reactive, draw the mechanism of its undergoing hydrolysis (reaction with H2O): Why makes this type of carbonyl so reactive to nucleophiles?arrow_forwardWhich solvents favor SN1 reactions and which favor SN2 reactions?arrow_forward
- Why a nitro group (NO2) directs electrophilic aromatic substitution tothe meta position ?arrow_forwardWhich of these alkyl halides do you expect to be most reactive in a reaction SN2 for a given nucleophile? Show the reason.arrow_forwarda. Compound X is benzene, Y is acetic anhydride acid. Complete the following scheme and determine Z! b. Determine which reagents except acetic acid anhydrides can replace Y!arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





