
Campbell Biology: Australian And New Zealand Edition + Mastering Biology With Etext
11th Edition
ISBN: 9781488687075
Author: Lisa, A. Urry
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 10TYU
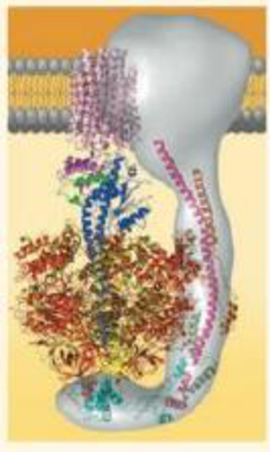
VISUAL SKILLS This Computer model shows the four parts of ATP synthase, each part consisting of a number of polypeptide subunits (the structure in gray is still an area of active research). Using Figure 9.14 as a guide, label the rotor, stator, internal rod, and catalytic knob of this molecular motor.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Graph B above depicts Lineweaver-Burk double reciprocal plot for an enzyme catalyzed reaction carried out in the presence or absence of an inhibitor. Which of the following statement best describes the kinetic data shown below: ?
Line 1 depicts the enzyme-catalyzed reaction carried out in the presence of a competitive inhibitor.
Line 1 depicts the enzyme-catalyzed reaction carried out in the presence of a noncompetitive inhibitor.
Line 2 depicts the enzyme-catalyzed reaction carried out in the presence of a competitive inhibitor.
Line 2 depicts the enzyme-catalyzed reaction carried out in the presence of a noncompetitive inhibitor.
ter reading the "Using spectrophotometers to study a Catechol Oxidase Reaction"
section, copy into your notebook the simplified catechol>benzoquinone equation
(include the molecule shapes, not just their names).
Biochemical events in the synthesis of ATP:
I. Subunits of ATP synthase chages in conformation and twists/rotates as H+ moves through it
II. H+ gradient is the driving force (through the ATP synthase) to form ATP from ADP + Pi.
III. H+ moving across the membrane through ATP synthase rotates its ring of c subunits
II and III only
I, II, III
I and II only
I and III only
Chapter 9 Solutions
Campbell Biology: Australian And New Zealand Edition + Mastering Biology With Etext
Ch. 9.1 - Compare and contrast aerobic and anaerobic...Ch. 9.1 - WHAT IF? If the following redox reaction...Ch. 9.2 - VISUAL SKILLS During the redox reaction in...Ch. 9.3 - Prob. 1CCCh. 9.3 - What process in your cells produce the CO2 that...Ch. 9.3 - VISUAL SKILLS The conversions shown in Figure...Ch. 9.4 - WHAT IF? What effect would an absence of O2 have...Ch. 9.4 - WHAT IF? In the absence of O2 as in question 1,...Ch. 9.4 - MAKE CONNECTIONS Membranes must be fluid to...Ch. 9.5 - Consider the NADH formed during glycolysis. What...
Ch. 9.5 - WHAT IF? A glucose-fed yeast cell is moved from...Ch. 9.6 - MAKE CONNECTIONS Compare the structure of a fat...Ch. 9.6 - Prob. 2CCCh. 9.6 - Prob. 3CCCh. 9.6 - VISUAL SKILLS During intense exercise, can a...Ch. 9 - Describe the difference between the two processes...Ch. 9 - Which reactions in glycolysis are the source of...Ch. 9 - What molecular products indicate the complete...Ch. 9 - Briefly explain the mechanism by which ATP...Ch. 9 - Prob. 9.5CRCh. 9 - Prob. 9.6CRCh. 9 - Level 1: Knowledge/Comprehension 1. The immediate...Ch. 9 - Prob. 2TYUCh. 9 - 3. The final electron acceptor of the electron...Ch. 9 - Prob. 4TYUCh. 9 - What is the oxidizing agent in the following...Ch. 9 - When electrons flow along the electron transport...Ch. 9 - Prob. 7TYUCh. 9 - Prob. 8TYUCh. 9 - MAKE CONNECTIONS The proton pump shown in Figures...Ch. 9 - VISUAL SKILLS This Computer model shows the four...Ch. 9 - INTERPRET THE DATA Phosphofructokinase is an...Ch. 9 - DRAW IT The graph here shows the pH difference...Ch. 9 - EVOLUTION CONNECTION AIP synthases are found in...Ch. 9 - SCIENTIFIC INQUIRY In the 1930s, some physicians...Ch. 9 - WRITE ABOUT A THEME: ORGANIZATION In a short essay...Ch. 9 - SYNTHESIZE YOUR KNOWLEDGE Coenzyme Q (CoQ) is sold...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- molecule A Plot of velocity versus substrate B Lineweaver-Burk plot 1/v Km 1 Vmax (S) Vmx 1 V max 1/2Vmax 1/Vmax -1/Km Km [S] 1/[S] fppt.com molecule Exercise The following data describe an enzyme-catalyzed reaction. Plot these results using the Lineweaver-Burk method, and determine values for KM and Vinax- The symbol mM represents millimoles per liter; 1 mM = 1 × 10 3 mol L. (The concentration of the enzyme is the same in all experiments.) Velocity (mM sec-) Substrate Concentration (тм) 2.5 0.024 5.0 0.036 10.0 0.053 15.0 0.060 20.0 0.061 fppt.comarrow_forwardImage shows reaction for hydrogen ion transport from outside mitochondrion to inside mitochondrion. Part a) What is ∆G ̊ for hydrogen ion transport. Part b) Outside the mitochondria there is a pH of 6.4 and inside mitochondria, there is a pH of 7.4. Calculate Q and ∆G for the reaction shown in the image. Part c) How many hydrogen ions needed per one ATP?arrow_forwardMAKE A GRAPH FOR ME ON GRAPH PAPER CALL IT ENZYMES VS RATE OF REACTION USING TABLE BELOW GRAPH paper INSERTED BELOW rules: data points must be an x or circled dot, must be on grid paper , the independant variable on the x axis and dependant variable on the y axis, must include titles Regarding the data points: - H2O2 + MnO2 Control #1: (Control #1, 5)- H2O2 + sand control #2: (Control #2, 0)- Plant versus Animal Liver Catalase: (Liver, 4)- Potato: Plant vs. Animal Catalase: (Potato, 3)- Substance Enzyme Concentration (Used Liver): (Liver Used, 4)- Substance Enzyme Concentration (Used H2O2): (Used H2O2, 1) - Boiling Water Bath Temperature: (Boiling Water Bath, 5)- Ice Water Bath Temperature: (Ice Water Bath, 2)- HCl, or pH 3: (H 3, 4)- NaOH at pH 12: (pH 12, 2)- pH 7 (H2O): (assuming average of pH readings; pH 7, not specified) The following explains how to display the graph: Title: Factors versus Enzyme Activity Rate - Labels on X- and Y-axes: Factors and Rate of Enzyme…arrow_forward
- LET'S APPLY 2: COMPUTATION - Directions: Compute the following questions. Write your solution and answer on your answer sheet. 1 GLUCOSE = 40 ATP MOLECULES (TOTAL) 1 GLUCOSE = 36 ATP MOLECULES (NET) 1 ATP MOLECULE = 7.3 kCal 1. How many ATP molecules will be produced when 100 molecules of glucose undergo cellular respiration? a. Total ATP produced: b. Total net ATP produced: 2. Compute for the amount of energy in kcal of 1a and 1b. a. Energy in kcal of the total ATP produced: b. Energy in kcal of the total net ATP produced:arrow_forwardRemember for T/F questions, either answer TRUE or FALSE, but if the answer is FALSE make sure to explain WHY the answer is false. The cell prefers lipids as a food source due to the large amount of NADH it can create during fatty acid oxidation is then used to create ATP. 1 A- Ff▼ = = F B I U S X₂ x² % S'S ↓ S 8- OiLearn Videoarrow_forwardVISUAL SKILLS During the redox reaction in glycolysis(see step 6 in Figure 9.9), which molecule acts as the oxidizing agent? The reducing agent?arrow_forward
- How do 3D model structure of transferase enzyme works? Give me a precise and accurate explanation. Thank you!arrow_forwardcell defense worksheet (1) [Compatibility Mode] Qv Search in Docume Insert Draw Design Layout References Mailings Review View Acrobat A. Century Gothic - 12 A A- U - abe X,2 x² A A Styles Styles Pane Create and Adobe PI Step 13: Take the "Energy and Transport Challenge!" How many ATP did you use? What type(s) of protein(s) were used? Explain when each type was used. Step 14: Take the "Osmosis Challenge!" What is Osmosis? What is the name of the special proieins that let water pass through? Is this passive or active transport? Step 15: From your Scores Sheet record: Lab Score (% correct): Number Correct: Numbar Incen et Page 2 of 3 * English (United States) 505 words E Focus MAR 15 MacBook Air 888 F1 F2 F3 F4 F5 F6 F7 F @ %23 $ & 7 8. Q W T Yarrow_forwardAutoSave 301-Enzyme Kinetics and Inhibition, Part 3 - Compatibility Mode - Word Search ff steve M SM File Home Insert Draw Design Layout References Mailings Review View Help E Share O Comments prepared five different concentrations of substrate (sucrose), and five different concentrations of inhibitor C (plus the control, with zero mM of inhibitor C). The following Table lists the inhibitor C concentrations [I], substrate concentrations [S], and resulting enzyme velocities (V.) for all six of these experiments: [I] O mM O mM O mM O mM O mM [S] Vo 1/[S] 1/ Vo 0.1 mM 0.333333333333 mM per minute 0.2 mM 0.50 0.3 mM 0.60 0.4 mM 0.666666666667 0.5 mM 0.714285714286 0.1 mM 0.1 mM 0.166666666667 0.1 mM 0.2 mM 0.25 0.1 mM 0.3 mM 0.30 0.1 mM 0.4 mM 0.333333333333 0.1 mM 0.5 mM 0.357142857143 0.20 mM 0.1 mM 0.111111111111 0.20 mM 0.2 mM 0.166666666667 0.20 mM 0.3 mM 0.20 0.20 mM 0.4 mM 0.222222222222 0.20 mM 0.5 mM 0.238095238095 0.3 mM 0.1 mM 0.083333333333 0.3 mM 0.2 mM 0.125 0.3 mM 0.3 mM…arrow_forward
- Based on the information in the data table and your graph, explain the relationship between percent concentration of catalase and rate of reaction. Using the graph, you created, predict the rate of the reaction if catalase concentrations are 10%, 60% and 85%. This will be done by extrapolating from the graph. In order to do this, take each catalase concentration above and locate it on the x-axis. From that point, draw a vertical line until it intersects the “best fit” line. From that point of intersection, draw a horizontal line to the y-axis. Where this intersection occurs would be the approximate rate of reaction.arrow_forwardMATHEMATICAL Using the data in Table 15.1, calculate the value of G for the following reaction. Creatinephosphate+GlycerolCreatine+Glycerol-3-phosphate Hint: This reaction proceeds in stages. ATP is formed in the first step, and the phosphate group is transferred from ATP to glycerol in the second step.arrow_forwardPlease answer fast The equilbrium constant (Keq) under standard conditions for the hydrolysis of ATP is 200,000 M. This would suggest the reaction is: 1. proceeding in reverse direction 2. proceeding in forward direction 3. in equilibrium However, this does not take into consideration the concentration of substrates/products within the cell. This is can be calculated by determining the reaction quotient, Q by: [ATP] x [Pi] / [ADP] [ATP] + [Pi] / [ADP] [ADP] x [ATP] / [Pi] [ADP] x [Pi] / [ATP] Biochemical reactions commonly involve the transfer of groups from ATP. What is one of the products of pyrophosphate cleavage from ATP? AMP Adenosine ADP Inorganic phosphate Q5-6. The hydrolysis of ATP is often coupled to other reactions. For example: Phosphoenolpyruvate (PEP) + H2O → Pyruvate + Pi (ΔG'° = -63.1 kJ/mol) ATP + H2O → ADP + Pi (ΔG'° = -30.5 kJ/mol) (The standard free-energy changes for the reactions are indicated in brackets) Q5. Given the information above, what is the…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning
Anaerobic Respiration; Author: Bozeman Science;https://www.youtube.com/watch?v=cDC29iBxb3w;License: Standard YouTube License, CC-BY