
Concept explainers
The compound butyric acid gives rancid butter its characteristic odor. (7.1, 7.2)
a. If black spheres are carbon atoms, white spheres are hydrogen atoms, and red spheres are oxygen atoms, what is the molecular formula of butyric acid?
b. What is the molar mass of butyric acid?
c. How many grams of butyric acid contain 3.28 x 1O atoms of oxygen?
d. How many grams of carbon are in 5.28 g of butyric acid?
e. Butyric acid has a density of 0.959 g/mL at 20 °C. How many moles of butyric acid are contained in 1.56 mL of butyric acid?
f. Identify the bonds C—-C, C- H, and C— 0 in a molecule of butyric acid as polar covalent or nonpolar covalent.
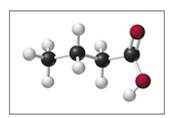
Butyrlc acid produces the characteristic odor of rancid butter.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Study Guide And Selected Solutions Manual For Chemistry Format: Paperback
- (5.8)Which of the following reactions will form a gaseous product? O H₂CO3(aq) + Pb(NO3)2(aq) O NaOH(aq) + HNO3(aq) O None of these O Na₂SO3(aq) + H₂SO4(aq) ◄ Previousarrow_forwardA student sets up and solves the following equation to solve a problem in solution stoichiometry. Fill in the missing part of the student's equation. (47.6) 65.08 mol 1 mL -3 10 L mol (3²) 3.2 L = 2.3 x 10 mL x10 ロ・ロ X μarrow_forward(3.9)Calculate the percent composition of sulfur in aluminum sulfate, Al2(SO4)3. 28.1% 17.7% 71.9% 33.6% ◄ Previousarrow_forward
- 20 cm3 of hydrogen peroxide solution of unknown concentration reacts completely with 12.15 cm3 0.04 mol/dm3 of potassium permanganate solution in sulfuric acid media. Calculate the mass concentration of hydrogen peroxide in g/dm3. How many cm3 (USC) of oxygen is formed? (2.07; 29.77)arrow_forward(3.6)What is the formula of the compound that forms between Pb4+ ion and sulfite ion? Pb₂SO3 O Pb2(SO3)4 O Pb(SO3)2 O Pb4(SO3)2 << Previousarrow_forwardhemistry-4th Hour - Dr. Paul / Gases/Lesson 148 Honor 22. According to Charles' law, what kind of relationship does temperature and volume have? O direct relationship O no relationship O indirect relationship chemical relationship GET IT ablo! (3.0.227)arrow_forward
- 7. What type of product is formed when acids are added to some ionic compounds? Stronger acids Weaker acids Gas Solids GET New version available! (3.0.220)arrow_forward5. (10 pts) Sulfuric acid (H2SO4) reacts with sodium hydroxide (NaOH) to produce water (H2O) and sodium sulfate (Na2SO4). In order to react 100 g of sulfuric acid, how many grams of sodium hydroxide are required?arrow_forward5) How many moles of sulfuric acid are present in 0.500 L of 0.150 M solution? (0.0750) 6) How many grams of sulfuric acid are in the above solution? (7.35)arrow_forward
- How many different chemical compounds are in the gas phase? 2. 3. 4.arrow_forward9. Which element is oxidized in the following reaction: 2FECI2 + Cl2 → 2FECI3 if Fe goes from +2 to +3 and CI goes from 0 to –2? Fe Neither element CI Cl and Fe GET IT NOW New version available! (3.0.220) PREVIOUSarrow_forwardP. How many moles of oxygen are needed to produce water when 4 x 10 molecules of hydrogen are used? (0.3). -> H,O H2 + O2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





