
GENETICS(LL)-W/CONNECT >CUSTOM<
6th Edition
ISBN: 9781260571561
Author: HARTWELL
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 16P
Suppose you are using a plasmid cloning vector that has no EcoRI sites (5′ G^AATTC 3′) in its polylinker because the particular drug resistance gene your vector contains has an EcoRI site within it.
| a. |
How could you use the following two oligonucleotides (and ligase enzyme) to ligate an insert that is an EcoRI fragment into the BamHI site (5′ G^GATCC 3′) in the polylinker of your vector?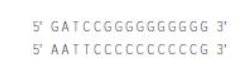 |
| b. | How many EcoRI sites will the recombinant DNA contain? How many BamHI sites? |
| c. | In part (a), you used the two oligonucleotides to make a so-called adapter. Adapters can also be used to ligate blunt-ended inserts into vectors cut with sticky-ended enzymes. Design an adapter that would allow you to ligate blunt-ended inserts into the BamHI site of your vector’s polylinker. (Note: Two blunt-ended DNA fragments can be ligated together, although the reaction is much less efficient than sticky-end ligation.) |
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1)Explain why there is a part of the Lacz gene in pUC18119 and also how the natural biochemical process wherein it normally functions is used during cloning by molecular biology.
2)Write down the basepairs of double-stranded ONA that is generated at the joint between a plasmid that is digested with Xhol and a DNA fragment that is digested with Sall and joined by DNA-ligase. Indicate the 5'- and 3'- termini of each strand.
A 2.0kb bacterial plasmid ‘BS1030’ is digested with the restriction endonuclease Sau3A; the plasmid map is depicted in the diagram below and the Sau3A (S) restriction sites are indicated.
Which of the following DNA fragments do you expect to see on an agarose gel when you run Sau3A-digested plasmid ‘BS1030’ DNA?
a.
250 bp, 450 bp, 550 bp, 1.1 kb, 1.5 kb and 2.0 kb
b.
2.0kb
c.
250 bp, 400 bp, 450 bp, 500 bp and 550 bp
d.
100 bp, 200 bp, 250 bp, 400 bp, 500 bp and 550 bp
After Drosophila DNA has been treated with a restriction enzyme, the fragments are inserted into plasmids and selected as clones in E. coli. With the use of this “shotgun” technique, every DNA sequence of Drosophila in a library can be recovered.a. How would you identify a clone that contains DNA encoding the protein actin, whose amino acid sequence is known?b. How would you identify a clone encoding a specific tRNA?
Chapter 9 Solutions
GENETICS(LL)-W/CONNECT >CUSTOM<
Ch. 9 - Match each of the terms in the left column to the...Ch. 9 - For each of the restriction enzymes listed below:...Ch. 9 - The calculations of the average restriction...Ch. 9 - The DNA molecule whose entire sequence follows is...Ch. 9 - Why do longer DNA molecules move more slowly than...Ch. 9 - Agarose gels with different average pore sizes are...Ch. 9 - The following picture shows the ethidium...Ch. 9 - The linear bacteriophage genomic DNA has at each...Ch. 9 - Consider a partial restriction digestion, in which...Ch. 9 - The text stated that molecular biologists have...
Ch. 9 - a. What is the purpose of molecular cloning? b....Ch. 9 - a. DNA polymerase b. RNA polymerase c. A...Ch. 9 - Is it possible that two different restriction...Ch. 9 - A plasmid vector pBS281 is cleaved by the enzyme...Ch. 9 - A recombinant DNA molecule is constructed using a...Ch. 9 - Suppose you are using a plasmid cloning vector...Ch. 9 - Prob. 17PCh. 9 - The lacZ gene from E. coli encodes the enzyme...Ch. 9 - Your undergraduate research advisor has assigned...Ch. 9 - Which of the enzymes from the following list would...Ch. 9 - You use the primer 5 GCCTCGAATCGGGTACC 3 to...Ch. 9 - a. To make a genomic library useful for sequencing...Ch. 9 - Problem 15 showed part of the sequence of the...Ch. 9 - Eukaryotic genomes are replete with repetitive...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Knowing that you are using HindIII and EcoRI to cut your plasmids, and that those two enzymes cut within the MCS, use the map of pUC19 provided below to compute: What will be the sizes of the 2 restriction fragments if NO insert is present in pUC19? What will be the sizes of the 2 restriction fragments if the approximately 317 bp RT-PCR product (insert from WT satC dimer) was ligated successfully into the SmaI site? What will be the sizes of the 2 restriction fragments if TWO approximately 317 bp RT-PCR products (2 ligated inserts from WT satC dimer) were ligated into the SmaI site?arrow_forwardExamine the structure of the pBR322 plasmid depicted below. Assume total size of the plasmid is 4,361 bp and the blue numbers indicate locations of restriction sites relative to the O point at the top of the plasmid. What size fragments would be generated by the following restriction digestion reactions? 1. Sall 2. Sal 1 + BamH1 3. Sal 1 + EcoR1 4. Sal I + BamH1 + EcoR1 PstI 3607 3000 4000 amp ori HindIII Edit View Insert Format Tools Table EcoRI EcoRV 4359 0 29 185 pBR322 4361 bp 2295 NdeI tet 2000 BamHI 375 651 SalI 1000 Type a short answer in the space provided below.arrow_forwardConsider the following plasmid (size 8000 bp), with restriction sites at the positions indicated: (see image) a) This plasmid is digested with the enzymes listed below. Indicate how many fragments will begenerated in each case, and give the sizes of the fragments.PstIXhoICombination of PstI + XhoI + EcoRI (triple digest) b) Draw the banding pattern you would expect to observe if each of these digestions is loaded into a separate well of an agarose gel, and the fragments separated by electrophoresis. In the first well you load a DNA marker (M) containing fragments with sizes of 1000 bp, 2000 bp, 4000 bp and 8000 bp. c) This gel is transferred to a membrane in a Southern blot experiment, and hybridised to a radioactively labelled 200 bp probe, which anneals to the plasmid DNA at the position indicated on the diagram above. Draw the autoradiographic profile you would expect to observe for the membrane.arrow_forward
- a)Dr. Thisisaneasyexam decides to amplify a gene from a plasmid using PCR. She starts out with 6.6 x 10-14g of a 10 kb template in a 100 µl reaction. Assuming that the molecular weight of the average base pair is 660 Daltons calculate the number of molecules of the template. b)Given that the concentration of each primer (20 base pairs each) is 0.1µM in this same reaction volume (100 µl) calculate the number of molecules of the primer presentarrow_forwardThe chain terminator method was used to sequence the following DNA fragment: ACTGGGCATAAGCGGGAACTTTGCAGAACTGGCTGGCCTCAGAGCAGGGA. 1. Predict a band pattern in a gel after sequencing this DNA fragment using a radioactively labeled primer [32P]-5’- TCTGAGGCCAGCCAGTTCTGCAAAGTTC. 2. Due to an experimental mistake, dATP was not added in all four reaction mixtures. How does the band pattern change?arrow_forwardYou want to make a recombinant DNA in which aPCR product amplified from the human genome is inserted into a plasmid vector. The polylinker of thisvector includes recognition sites for the enzymesEcoRI (5′ G^AATTC 3′) and BamHI (5′ G^GATCC3′). (The ^ symbolizes the cut site in the DNA.) PCRprimers that could amplify the fragment of humanDNA are: 5′ GCTACTTCGCGTATTCCA 3′ and5′ CCCAAGTCCTAGCCGATA 3′.a. Describe in detail how these primers would need tobe modified to create a fragment of the human genome flanked by EcoRI sticky ends so that thisfragment could be cloned easily into the plasmidvector. You will need to consider the fact that mostrestriction enzymes, including EcoRI, cannot cutDNA if the restriction site is directly at the end ofthe DNA molecule; the restriction enzyme recognition site must be at least six base pairs distant fromthe end.b. Describe a potential feature of the PCR-amplifiedregion of the human genome that could prevent youfrom using the strategy you described in…arrow_forward
- You are attempting to clone a 3 kb gene from the bacterial sp Microbacterium foliarum into the EcoRI site of the 6.0 kb plasmid shown. If you restrict the plasmid with EcoR1, how many bands will you obtain on agarose gel electrophoresis? Compare it with a plasmid in which no gene has been inserted. Draw a representative agarose gel showing the bands, and the direction of current flow as well point out the positive and negative electrodesarrow_forwardSuppose that you want to sequence the following DNA fragment: 5′–TCCCGGGAAA-primer site–3′ You first use PCR to amplify the fragment, so that there is sufficient DNA for sequencing. You carry out dideoxy sequencing and then separate the products of the sequencing reactions by gel electrophoresis. Draw the bands that should appear on the gel from the four sequencing reactions.arrow_forwardClone number in this case is number 196 which is shown in the images. State whether a BamHI site has been re-created at the forward- and the reverse-end junctions of the human DNA with the plasmid vector band sizes are shown in one of the images. (0.5, 1, 2, 3, 4, 5, 6, 8, 10kb)arrow_forward
- Human genomic libraries used for DNA sequencing are often made from fragments obtained by cleaving human DNA with Haeiii in such a way that the DNA is only partially digested; that is, not all the possible HaeIII sites have been cleaved. What is a possible reason for doing this?arrow_forwardWhen joining two or more DNA fragments, a researcher can adjust the sequence at the junction in a variety of subtle ways, as seen in the following exercises.(a) Draw the structure of each end of a linear DNA fragment produced by an EcoRI restriction digest (include those sequences remaining from the EcoRI recognition sequence).(b) Draw the structure resulting from the reaction of this end sequence with DNA polymerase I and the four deoxynucleoside triphosphates.(c) Draw the sequence produced at the junction that arises if two ends with the structure derived in (b) are ligated (d) Draw the structure produced if the structure derived in (a) is treated with a nuclease that degrades only single-stranded DNA.(e) Draw the sequence of the junction produced if an end with structure (b) is ligated to an end with structure (d).(f) Draw the structure of the end of a linear DNA fragment that was produced by a PvuII restriction digest (include those sequences remaining from the PvuII recognition…arrow_forwardWhich of the following set(s) of primers a-d could you use to amplify the following target DNA sequence, which is part of the last protein-coding exon of the CFTR gene? Explain briefly. (Note: The three dots represent the body of the region to be amplified, whose beginning and end are only being shown.) 5' GGCTAAGATCTGAATTTTCCGAG . TTGGGCAATAATGTAGCGCCTT 3' 3' CCGATTCTAGACTTAAAAGGCTC . AACCCGTTATTACATCGCGGAA 5' a. 5' GGAAAATTCAGATCTTAG 3'; 5' TGGGCAATAATGTAGCGC 3' b. 5' GCTAAGATCTGAATTTTC 3'; 3' ACCCGTTATTACATCGCG 5' c. 3' GATTCTAGACTTAAAGGC 5'; 3' АССCGTTATTАСАТСGCG 5 d. 5' GCTAAGATCTGAATTTTC 3'; 5' TGGGCAATAATGTAGCGC 3'arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Bacterial Genomics and Metagenomics; Author: Quadram Institute;https://www.youtube.com/watch?v=_6IdVTAFXoU;License: Standard youtube license