
Concept explainers
Compound S
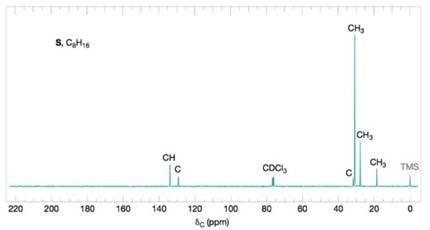
Figure 9.38 The broadband proton-decoupled
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
EBK ORGANIC CHEMISTRY
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
General Chemistry: Atoms First
Fundamentals of Heat and Mass Transfer
Chemistry: A Molecular Approach (4th Edition)
Chemistry & Chemical Reactivity
Elementary Principles of Chemical Processes, Binder Ready Version
- The following 1H NMR peaks were recorded on a spectrometer operating at 200 MHz. Convert each into δ units. (a) CHCl3; 1454 Hz (b) CH3Cl; 610 Hz (c) CH3OH; 693 Hz (d) CH2Cl2; 1060 Hzarrow_forwardFollowing is the mass spectrum of an unknown compound. The two highest peaks are at m/z 120 and 122. Suggest a structure for this compound. (Data from http://webbook.nist.gov/chemistry/.)arrow_forwardHow many peaks would you expect to see in the 13C NMR spectrum of this substance?arrow_forward
- Complete the spectroscopy data tables for a compound with molecular formula C6H12O2. Determine the structure of the compound. Any labile protons, if they exist, will not be present in this particular 1H NMR spectrum.arrow_forwardThe mass spectrum of 2,3-dimethylpentane also shows peaks at mlz = 57 and 43. Propose possible structures for the ions that give rise to these peaks.arrow_forward4. A compound having the molecular formula CsH10O2 exhibits the following 'H-NMR-Figure 4a Figure 4a Figure 4b Multiplet @ 1.46 ppm Multiplet @ 1.22 ppm Triplet @ 2.20 ppm Triplet @ 0.78 ppm PPM and 13C-NMR-Figure 4b spectra. Identify the compound. The spin multiplicities are indicated below. 'H-NMR (400 MHz, CDCI3 at 300K) 8: 11.8 (s), 2.20 (t), 1.46 (m), 1.22 (m), 0.78 (t). 13C-NMR (400 MHz, CDCI3 at 300K) 8: 180.8 (s), 33.8 (t), 26.8 (t), 22.4 (t), 3.58 (q).arrow_forward
- What will be the 1H-NMR spectrum for the next molecule BrClCHCH2COCH3 and illustrate the corresponding signals according to their chemical displacement, multiplicity and integration. Indicate two basic differences that exist between the spectra of 1H y 13C in NMR. Establish the difference between the multiplicity of a signal and the integral (integration) of the signal in spectroscopy of 1H-NMR.arrow_forwardORGANIC CHEMISTRY: SEE PICTURE A compound with the chemical formula C5H10O gives the H NMR spectra below. Determine the HDI and then propose a structure of the unknown. Annotate the spectra with all relevant signals identified to support your answer. Hint: both peaks below could be classified as a singlet.arrow_forwardThe 1H-NMR spectrum of the compound with the closed formula C8H16O2 is given below. Find the possible structure of the compound by analyzing the spectrum.arrow_forward
- How many peaks would the 1H NMR spectrum of the following molecule have?arrow_forwardb. Predict the structure of a compound based on this 18C NMR spectra. The chemical formula of this compound is C4H&O2. Briefly explain your answer. 200 180 160 140 120 100 80 60 40 20 8 ppmarrow_forward300 MHz 'H NMR spectra in CDCI3 are shown below for five of the six isomers of dimethylphenol. Match each spectrum to the appropriate compound and assign the resonances of the spectrum to the appropriate protons in the compoundarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning


