
EBK ORGANIC CHEMISTRY
12th Edition
ISBN: 9781119233664
Author: Snyder
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 3PP
PRACTICE PROBLEM 9.3
Using the method of Section 9.5A, determine the number of expected signals for the following compounds.
(a)
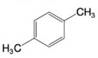
(b)
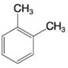
(c)
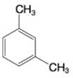
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the characteristic infrared absorptions of the functional groups in the following molecules.
(f) pentanoic acid (g) pentanenitrile
Predict the characteristic infrared absorptions of the functional groups in the following molecules.
(a) ethyl acetate (b) pentanamide
Нас
3. (5S,7R) -7-bromo-3,9-diethyl-6,6-dimethylundec-3,8-dien-5-ol
H C
Н.С
Ay statuvos
OH
CH3
Ay si
Br
Н
CH3
CH3
I
b. number of peaks =
С.
Chapter 9 Solutions
EBK ORGANIC CHEMISTRY
Ch. 9 - Prob. 1PPCh. 9 - PRACTICE PROBLEM 9.2 What compound with molecular...Ch. 9 - PRACTICE PROBLEM 9.3
Using the method of Section...Ch. 9 - PRACTICE PROBLEM 9.4 How many signals would each...Ch. 9 - Prob. 5PPCh. 9 - Prob. 6PPCh. 9 - PRACTICE PROBLEM 9.7
The relative chemical shifts...Ch. 9 - Prob. 8PPCh. 9 - PRACTICE PROBLEM 9.9 Propose a structure for...Ch. 9 - PRACTICE PROBLEM 9.10
What is the dihedral angle...
Ch. 9 - PRACTICE PROBLEM 9.11 Draw the most stable chair...Ch. 9 - Prob. 12PPCh. 9 - PRACTICE PROBLEM 9.13 How many signals would you...Ch. 9 - Prob. 14PPCh. 9 - Prob. 15PPCh. 9 - Prob. 16PPCh. 9 - Prob. 17PPCh. 9 - PRACTICE PROBLEM 9.18
What are the expected ratios...Ch. 9 - Prob. 19PPCh. 9 - How many 1H NMR signals (not peaks) would you...Ch. 9 - How many 13C NMR signals would you predict for...Ch. 9 - Prob. 22PCh. 9 - Prob. 23PCh. 9 - Prob. 24PCh. 9 - Compound Q has the molecular formula C7H8. The...Ch. 9 - 9.26 Explain in detail how you would distinguish...Ch. 9 - Compound S (C8H16) reacts with one mole of bromine...Ch. 9 - A compound with molecular formula C4H8O has a...Ch. 9 - In the mass spectrum of 2, 6-dimethyl-4-heptanol...Ch. 9 - Prob. 30PCh. 9 - What are the masses and structures of the ions...Ch. 9 - Prob. 32PCh. 9 - Ethyl bromide and methoxybenzene (shown below)...Ch. 9 - 9.34 The homologous series of primary amines, ,...Ch. 9 - Propose a structure that is consistent with each...Ch. 9 - 9.36 Propose structures for compounds E and F....Ch. 9 - 9.37 Use the NMR and IR data below to propose a...Ch. 9 - 9.38 When dissolved in , a compound (K) with the...Ch. 9 - Compound T (C5H8O) has a strong IR absorption band...Ch. 9 - Prob. 40PCh. 9 - Deduce the structure of the compound that gives...Ch. 9 - Deduce the structure of the compound that gives...Ch. 9 - The 1H NMR spectrum of a solution of 1,...Ch. 9 - Acetic acid has a mass spectrum showing a...Ch. 9 - The 1H NMR peak for the hydroxyl proton of...Ch. 9 - The 1H NMR study of DMF (N, N-dimethylformamide)...Ch. 9 - 9.48 The mass spectra of many benzene derivatives...Ch. 9 - Prob. 49PCh. 9 - 1. Given the following information, elucidate the...Ch. 9 - Two compounds with the molecular formula C5H10O...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Derive Equation 6.54 for the boiling-point elevation of a dilute solution of a nonvolatile solute with mole fra...
Elementary Principles of Chemical Processes, Binder Ready Version
23. Give the symbol and name for (a) an isotope with a mass number of 37 and an atomic number of 17 and (b) an ...
Chemistry For Changing Times (14th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
The reaction of tert-butyl chloride with methanol (CH3)3CCl + CH3OH (CH3)3COCH3 + HCl tert butyl chloride met...
Organic Chemistry (9th Edition)
3.1 The reaction between reactant A (blue spheres) and reactant B (red spheres) is shown in the
following diag...
Chemistry: The Central Science (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The compound whose 1H NMR spectrum is shown has the molecular formula C7H7Br. Follow the following questions to predict the unknown structure. 2(a) Degree of the unsaturation of this compound is= , 2(b) The two distinct peaks in the aromatic region of the 1H NMR indicate that compound is .................-disubstituted = 2(c) The splitting pattern of the peak at 2.31 δ is = 2(d) The group that corresponds to the splitting pattern in 1(c) is = 2(e) This compound has the plane of symmetry Yes or No = 2(f) The name of the unknown compound =arrow_forwardyou infrared spectroscopy. (a) (b) H distinguish between the following pairs of compounds by using NH2arrow_forwardDraw the structural formulas of the following compounds and indicate the number of NMR signals that would be expected for each compound. (a) methyl iodide (b) 2,4-dimethylpentane (c) cyclopentane (d) propylene (propene)arrow_forward
- You are given two compounds, dimethylamine (CH3)2NH and ethanamide, CH3CONH2. a) b) c) Which compound is more basic? Explain your answer. How do you distinguish them by infrared spectroscopy? Outline an extraction method to separate a mixture of these two compounds.arrow_forwardDescribe the characteristic infrared absorption frequencies that would allow you to distinguish between the following pairsof compounds.(a) 2,3-dimethylbut-2-ene and 2,3-dimethylbut-1-enearrow_forwardConsider the following three compounds: CH,=CH, CH,=CH-CH=CH, CH,=CH-CH=CH-CH=CH, (1) (2) (3) Compound 1 contains a simple isolated carbon-carbon double bond, but the other two have conjugated double bonds. (i) Which compound will have the largest absorption (max) Value? (ii) Give a reason for your answer in part (b)(i).arrow_forward
- Perform symmetry analysis on the compounds below and predict the number of ¹3C NMR signals expected for each compound. Then predict how many signals would be in the following chemical shift ranges: 0-50, 50-100, 100-160, or 160-220 ppm for each compound. (b) للہ (c) (d) LOHarrow_forwardPredict the theoretical number of different NMR signals produced by each compound, and give approximate chemical shifts. Point out any diastereotopic relationships. (a) Ph¬CHBr¬CH2Br (b) vinyl chloridearrow_forwardPredict the characteristic infrared absorptions of the functional groups in the following molecules. (a) cyclohexene (b) pentan-2-ol (c) pentan-2-onearrow_forward
- Which compound will give a broad and intense band at 3100 cm-¹ in its IR spectra? (a) Pentane (c) Pentanol Pentanone Propyl ethanoatearrow_forwardDistinguish the characteristics infrared absorption frequencies that would allow you to distinguish the following pair of compounds. (Picture attached)arrow_forward(a) How many proton and carbon signals are expected for the 2 possible regioisomers? Clearly mark the chemically distinct/inequivalent carbons in your structures. (b) Estimate the 1H chemical shift for HB of 2-indanol and HA of 1-indanol. Is one more downfield than the other or are they expected to be similar? Explain your answer.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY