
(a)
Interpretation:
The vinyl carbocations formed when phenyl-substituted acetylenes are protonated and the regioselectivity observed in the reaction have to be drawn and explained.
Concept Introduction:
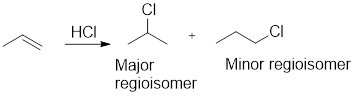
Regioselectivity: It is the favouring of reactants or reagents to bond to one atom or another. Regioisomers are isomers in which connectivity of atoms varies but same numbers of atoms are present in it. An example of reaction between propene and hydrochloric acid for regioisomers is given as,
(b)
Interpretation:
By relating the stabilities of transition states of vinyl carbonations, the stereo selectivity in reactions of
Concept Introduction:
Stereoselectivity: A stereoselective reaction where only one stereoisomer reacts among mixture of stereoisomers reacts. This reaction gives two or more stereoisomers but produces almost only one stereoisomer product.
Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Organic Chemistry Third Edition + Electronic Solutions Manual And Study Guide
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





