
Custom eBook for Organic Chemistry
2nd Edition
ISBN: 9798214171104
Author: Straumanis
Publisher: Cengage Custom
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 9, Problem 8CTQ
Interpretation Introduction
Interpretation: The product for the given reaction should be predicted.
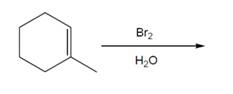
Concept introduction:
The reaction of an
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Why do you suppose this mechanism leads to the anti-Markovnikov product? Provide a rational and discuss the potential intermediates that are involved.
Which substrate will result in an alkene that could be consider both the Hoffman and Saytzev product?
The same reaction with chlorine is not much regioselective in the above. Explain the decreased regioselectivity of the following chlorination reaction by comparing the bromination reaction in the above (draw the mechanism, transition state, energy diagram, etc.). ‘Markovnikov’s or anti-Markovnikov’s rule’ is not a correct answer.
Chapter 9 Solutions
Custom eBook for Organic Chemistry
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Given three equally branched structures and with only a difference in the type of group attached to each (OH, NH3, or OTs), which compound would react faster in an SN1 reaction?arrow_forwardThe ratio between the products resulting from the reversible Diels-Alder reaction of furan and N- phenyl maleimide can be tuned by varying the reaction temperature and solvent. Please answer the following questions. A) Draw the products of this reaction that agree with the product ratios at each temperature when run either neat or in ether. Label each product as either endo or exo and be sure to indicate stereochemistry. Q furan n-phenyl maleimide + Time 7 days 7 days 20 days 7 days then 5 h Temp 0 °C ambient ambient 0 °C 60 °C A B 36 49 48 41 68a 23a 43 21 A + N-phenyl- maleimide (%) 14 11 ga 36 B) Draw the reaction coordinate diagram of this reaction. Label the products from part A, and provide a drawing of any transition states. Barrow_forwardProvide specific examples/reaction scheme/mechanism for each of the following: a) Hoffman elimination b) Wittig reaction c) Zaitsev's rule d) Anti-Markovnikov's rule e) Stereoelectronic effectarrow_forward
- Which reaction or statement regarding nucleophilic substitutions is incorrect? A) C₁ + 2 H2O ноттон + 2 HCI B) ta CI+MeOH to + HCI C) D) The rate-limiting step in SN1 reactions is the initial step, loss of the leaving group. Nucleophilic substitution reactions that follow second-order kinetics involve complete inversion of configuration.arrow_forwardFor the given molecule, highlight any nucleophilic sites in red and any electrophilic sites in blue. O: 439 0-Harrow_forwardFrom the below reactions, indicate which product is the Markovnikov product and which product is the anti-Markovnikov product?arrow_forward
- How do you properly write an SN1 reaction, with transition states/intermediate steps included?arrow_forwardAccording to the LUMO density map, on which face is the LUMO electronically more exposed to nucleophiles? Based on this data, which face would be more electronically favored? (These questions go hand in hand.)arrow_forwardPredict the Robinson annulation product.arrow_forward
- For the reagent sequence shown, what is the expected major product?arrow_forwardExplain the stereoselecticity of this reactionarrow_forwardWhen the molecule reacting with the electrophile is a strong nucleophile and a strong base, the substitution reaction is favoured when There is the least amount of crowding (steric hindrance) around the alpha carbon There is the most amount of crowding (steric hindrance) around the alpha carbon The leaving group is a strong basearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning