
Interpretation:
The complete and detailed mechanisms for the
Concept introduction:
Answer to Problem 9.1P
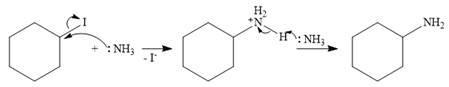
The mechanism for SN2 reaction:
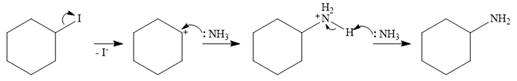
The mechanism for SN1 reaction:
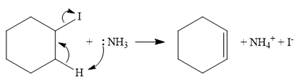
The mechanism for E2 reaction:
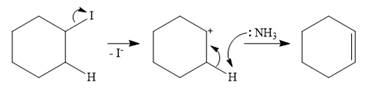
The mechanism for E1 reaction:
Explanation of Solution
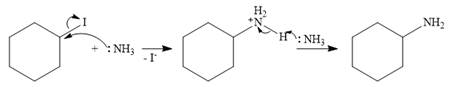
In the case of an SN2 mechanism, the nucleophile attacks the reactant, and at the same time, the leaving group departs. So the SN2 mechanism is a one-step reaction. As the nucleophile ammonia is neutral, an unstable charged species is formed.
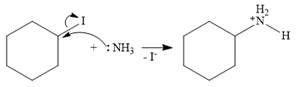
The base, another molecule of ammonia, extracts a proton from the charged species to form the stable, final product.
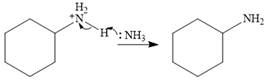
Therefore, the complete mechanism can be shown as below:
An SN1 mechanism is a two-step mechanism. In the first step, the leaving group departs along with the electron pair from its bond with the carbon atom. This generates a carbocation.
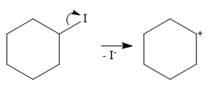
In the second step, the nucleophile ammonia uses the lone pair on nitrogen to form a bond with the carbocation to form a charged species.
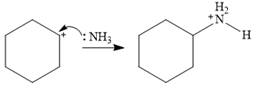
In the final step, another molecule of ammonia acts as a base and extracts a proton from the
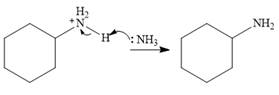
Therefore, the complete mechanism can be shown as below:

In the case of an E2 mechanism, ammonia acts as a base and abstracts a proton from a carbon atom adjacent to the one attached to the leaving group. The
Therefore, the complete mechanism can be shown as below:
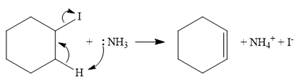
In the case of an E1 mechanism, the leaving group iodine departs with its bond pair, as an iodide anion. This forms the carbocation.
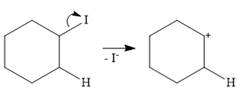
In the second step, ammonia acts as a base and extracts a proton from a carbon adjacent to the positively charged carbon. The
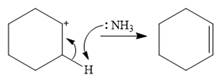
Therefore, the complete mechanism for the E1 reaction can be shown as below:
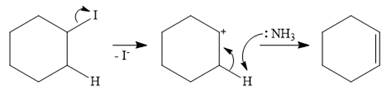
In a nucleophilic substitution reaction, the attacking species acts as a nucleophile while in an elimination reaction, it acts as a base.
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Draw this compound in a chair form so that an E2 reaction is possible. Then, draw the mechanism and the major product. Your solution and structures must clearly show the 3D nature of the E2 mechanism.arrow_forwardis this an E1 or E2 mechanism for this reaction? What is the major product and mechanism for it?arrow_forwardSelect one activator & ortho para director to be on a benzene ring to start. You will then add a bromine to this group showing the COMPLETE mechanism, including all resonance structures. Put a box around the stabilizing resonance structure and explain why it is stabilizing.arrow_forward
- Draw the MAJOR product(s) of this reactions:arrow_forwardDraw the complete mechanism for the reaction below, including stereochemistry.arrow_forwardFor each set of reactions, circle the mechanism (SN2 vs SN1), draw the main organic substitution/elimination product (for each reaction draw the product, though in some cases it may be equivalent) and indicate which reaction occurs at the faster rate.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
