
Modified Masteringchemistry With Pearson Etext -- Valuepack Access Card -- For Fundamentals Of General, Organic, And Biological Chemistry
8th Edition
ISBN: 9780134261270
Author: John E.; Ballantine, David S.; Hoeger, Carl A.; Peterson, Virginia E. Mcmurry
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 9, Problem 9.31UKC
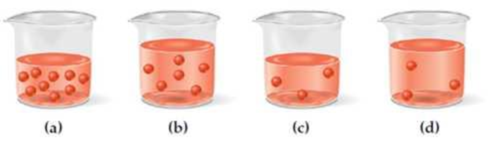
A beaker containing 150.0 mL of 0.1 M glucose is represented by (a). Which of the drawings (b)–(d) represents the solution that results when 50.0 mL is withdrawn from (a) and then diluted by a factor of 4?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the final concentration if 239mL of a 3.9M glucose solution is diluted to a volume of 842mL?
How many mL of the 1 M glucose stock solution do you need to prepare 100 mL of a 1 mM glucose solution?
How many milliliters of 0.0050 N KOH are required to neutralize 25 mL of 0.0050 N H2SO4? To neutralize 25 mL of 0.0050 M H2SO4?
Chapter 9 Solutions
Modified Masteringchemistry With Pearson Etext -- Valuepack Access Card -- For Fundamentals Of General, Organic, And Biological Chemistry
Ch. 9.1 - Classify the following liquid mixtures as...Ch. 9.2 - Write the formula of sodium sulfate decahydrate,...Ch. 9.2 - What mass of Glaubers salt must be used to provide...Ch. 9.2 - Which of the following pairs of substances would...Ch. 9.4 - A solution is prepared by dissolving 12.5 g of KBr...Ch. 9.5 - Prob. 9.4PCh. 9.5 - Prob. 9.5PCh. 9.6 - How does the body increase oxygen availability at...Ch. 9.6 - A metal alloy contains 15.8% nickel (m/m)%. What...Ch. 9.6 - Prob. 9.7P
Ch. 9.6 - Prob. 9.8PCh. 9.6 - Prob. 9.9PCh. 9.6 - Prob. 9.10PCh. 9.6 - Prob. 9.11PCh. 9.6 - What is the molarity of a solution that contains...Ch. 9.6 - How many moles of solute are present in the...Ch. 9.6 - The concentration of cholesterol (C27H46O) in...Ch. 9.6 - Prob. 9.15PCh. 9.7 - Prob. 9.16PCh. 9.7 - Prob. 9.17PCh. 9.8 - Prob. 9.5CIAPCh. 9.8 - Prob. 9.6CIAPCh. 9.8 - Prob. 9.7CIAPCh. 9.8 - Prob. 9.18PCh. 9.8 - Prob. 9.19PCh. 9.9 - A solution is prepared by dissolving 0.67 mol of...Ch. 9.9 - Prob. 9.21PCh. 9.9 - Prob. 9.22KCPCh. 9.9 - What is the freezing point of a solution of 1.0...Ch. 9.9 - Prob. 9.24PCh. 9.10 - What is the osmolarity of the following solutions?...Ch. 9.10 - Prob. 9.26PCh. 9.11 - Prob. 9.8CIAPCh. 9.11 - Prob. 9.9CIAPCh. 9 - Assume that two liquids are separated by a...Ch. 9 - Prob. 9.28UKCCh. 9 - Prob. 9.29UKCCh. 9 - Assume that you have two full beakers, one...Ch. 9 - A beaker containing 150.0 mL of 0.1 M glucose is...Ch. 9 - Prob. 9.32APCh. 9 - Prob. 9.33APCh. 9 - Prob. 9.34APCh. 9 - Prob. 9.35APCh. 9 - Which of the following are solutions? (a)Italian...Ch. 9 - Prob. 9.37APCh. 9 - The solubility of NH3 gas in water at an NH3...Ch. 9 - Prob. 9.39APCh. 9 - Is a solution highly concentrated if it is...Ch. 9 - How is mass/volume percent concentration defined...Ch. 9 - Prob. 9.42APCh. 9 - Prob. 9.43APCh. 9 - A 750.0 mL bottle of Listerine is of a 21% (v/v)...Ch. 9 - Prob. 9.45APCh. 9 - Prob. 9.46APCh. 9 - Prob. 9.47APCh. 9 - Prob. 9.48APCh. 9 - Prob. 9.49APCh. 9 - How many moles of each substance are needed to...Ch. 9 - Prob. 9.51APCh. 9 - If you had only 23 g of KOH remaining in a bottle,...Ch. 9 - Prob. 9.53APCh. 9 - The lethal dosage of potassium cyanide (KCN) in...Ch. 9 - What is the molarity of the following solutions?...Ch. 9 - How many grams of solute are in the following...Ch. 9 - How many milliliters of a 0.75 M HCl solution do...Ch. 9 - Prob. 9.58APCh. 9 - Prob. 9.59APCh. 9 - Sodium thiosulfate (Na2S2O3), the major component...Ch. 9 - Prob. 9.61APCh. 9 - What is the final volume of NaOH solution prepared...Ch. 9 - Prob. 9.63APCh. 9 - What is the concentration of a NaCl solution, in...Ch. 9 - Concentrated (12.0 M) hydrochloric acid is sold...Ch. 9 - Dilute solutions of NaHCO3 are sometimes used in...Ch. 9 - Prob. 9.67APCh. 9 - Prob. 9.68APCh. 9 - Prob. 9.69APCh. 9 - What is the total anion concentration (in mEq/L)...Ch. 9 - Kaochlor, a 10% (m/v) KCl solution, is an oral...Ch. 9 - Prob. 9.72APCh. 9 - The concentration of Cl ion in blood is...Ch. 9 - Prob. 9.74APCh. 9 - Which lowers the freezing point of 2.0 kg of water...Ch. 9 - Which solution has the higher boiling point, 0.500...Ch. 9 - Methanol, CH3OH, is sometimes used as an...Ch. 9 - Hard candy is prepared by dissolving pure sugar...Ch. 9 - Prob. 9.79APCh. 9 - Prob. 9.80APCh. 9 - Which of the following solutions has the higher...Ch. 9 - Which of the following solutions will give rise to...Ch. 9 - A pickling solution for preserving food is...Ch. 9 - Prob. 9.84APCh. 9 - Prob. 9.85CPCh. 9 - Emergency treatment of cardiac arrest victims...Ch. 9 - Nitric acid, HNO3, is available commercially at a...Ch. 9 - Prob. 9.88CPCh. 9 - A typical dosage of statin drugs for the treatment...Ch. 9 - Prob. 9.90CPCh. 9 - Prob. 9.91CPCh. 9 - Prob. 9.92CPCh. 9 - Prob. 9.93CPCh. 9 - Prob. 9.94CPCh. 9 - Prob. 9.95CPCh. 9 - Prob. 9.96GPCh. 9 - Look up the maximum concentrations set by the U.S....Ch. 9 - Look up the composition of Ringers solution used...Ch. 9 - To prevent accumulation of ice on roads and...Ch. 9 - Many carbonate minerals are insoluble in water and...Ch. 9 - Prob. 9.101GP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- How many grams of glucose (C6H2O6 molecular mass =180daltons) would be present in one liter of a 1M (molar) solution of glucose?arrow_forwardIn an experiment, what happens to the calculated molar mass after waiting an hour between thawing the frozen cyclohexane and adding the unknown compound, during which time some of the cyclohexane evaporated. Will the molar mass be too large, too small, or unaffected? Explain.|arrow_forwardYou have 10 mg/ml ethidium bromide solution. If you add 5 µl of this to 50 ml of agarose gel solution, what will be the final concentration of ethidium bromide in the gel solution in mg/ml?arrow_forward
- To make up a solution of phosphate buffered saline (PBS), you need 10 mM Na2HPO4 (anhydrous) (FW: 141.96 g/mol), 3 M NaCl (FW: 58.44 g/mol), and 5mM KH2PO4 (FW: 136.09 g/mol). How many grams of each will you need to make up 850 mL of PBS?arrow_forwardPropanamide and methyl acetate have about the same molar mass, both are quite soluble in water, and yet the boiling point of propanamide is 486 K, whereas that of methyl acetate is 330 K. Explain.arrow_forwardIf 150ml of polyethylene glycol 400 weighs 74.1g, what is the specific gravity?arrow_forward
- In order to make a solution of the concentration 0.364 M, what mass of NaCl should bedissolved in 152 mL of water?arrow_forwardHow many grams of NaOH are needed to make 50 mL of a 0.02 g/mL NaOH solution? How many grams of HCl are needed to make 10 uL of a 0.005g/mL acid solution? How many grams of CuSO4 would you use to make 1 L of an 8 g/L CuSO4 solution? 12 grams of glucose will make how many mL of a 0.5 g/mL solution? If you mix 9 g of NaOH with 90 mL of water, what is the mass/volume (g/mL) concentration of the solution?arrow_forwardIf you added three proteins (Protein A, pI 5.0; Protein B, pI 6.0; Protein C, pI 7.0) to an anion exchange column equilibrated with bicine buffer, pH 8.5, in what order would these protein be expected to elute as the column was loaded, washed, and then increasing amounts of NaCl were added?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you


GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license