
Concept explainers
Interpretation:
The three resonance structures for the given molecule
Concept Introduction:
Octet rule: According to octet rule, when an atom contains eight electrons in its valence shell (outermost shell), it will be stable. Most of the atom will donate or accept electrons to its outer most orbital to satisfy the octet rule.
The Lewis structure is based on the concept of the octet rule so that the electrons shared in each atom should have 8 electrons in its outer shell.
Sometimes the
All the possible resonance structures are imaginary whereas the resonance hybrid is real.
Resonance is a method used to describe about delocalized electrons inside certain molecules or polyatomic ions since the Lewis structure can’t express it. A molecule or ion containing delocalized electrons can be represented by using several similar structures such structures are called as resonance structures or canonical structures.
The general rules to be followed for drawing resonance structures are given below:
- 1. The position of the atoms does not change only the pi electrons and a lone pair of electrons can change their positions.
- 2. The total number of valence electrons in all the resonance structures remains the same.
- 3. Octet rule must not be violated. No atom can have electrons more than 8 in its valence shell except sulphur.
- 4. Transfer of electrons between the bonds is shown by curved arrows.
Formal charge of an atom can be determined by the given formula.
Answer to Problem 9.54QP
The resonance structure can be drawn as,
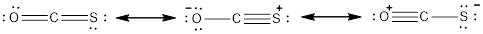
Explanation of Solution
Total electrons in
Accordingly Lewis structure of
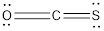
The resonance structures of molecule is as follows,
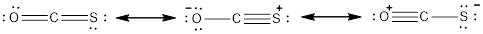
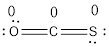
- Oxygen atom
Substituting these values to the equation,
- Carbon atom
Substituting these values to the equation,
- Sulphur atom
Substituting these values to the equation,
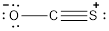
- Oxygen atom
Substituting these values to the equation,
- Carbon atom
Substituting these values to the equation,
- Sulphur atom
Substituting these values to the equation,
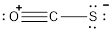
- Oxygen atom
Substituting these values to the equation,
- Carbon atom
Substituting these values to the equation,
- Sulphur atom
Substituting these values to the equation,
The three resonance structures for the given molecule
Want to see more full solutions like this?
Chapter 9 Solutions
General Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





