
Concept explainers
(a)
Interpretation:
For the given substrate, when
Concept introduction:
As the number of alkyl groups on the carbon bonded to the leaving group increases, the
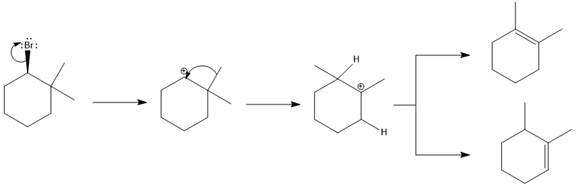
Answer to Problem 9.69P
The major E1 product for the given reaction is shown below:
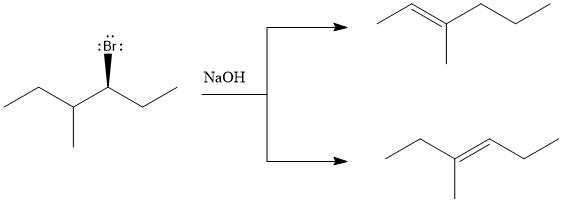
Explanation of Solution
Elimination usually takes place so as to produce the most stable, i.e., highly alkyl substituted, alkene.
The possible E1 products are obtained by eliminating the leaving group and a proton on carbon adjacent to the one bonded to the leaving group. Here the
The first and second alkene product is highly substituted than the third alkene product. Therefore, the first and second alkene products are more stable. The base in this reaction is OH, which is not bulky, so the first and second products are the major products.

From the stability of the product formed, the major E1 product is drawn.
(b)
Interpretation:
For the given substrate, when
Concept introduction:
As the number of alkyl groups on the carbon bonded to the leaving group increases, the rate of E1 reaction increases. Each additional alkyl group, which is electron donating, stabilizes the carbocation intermediate produced and thus helps the leaving group leave. The
Answer to Problem 9.69P
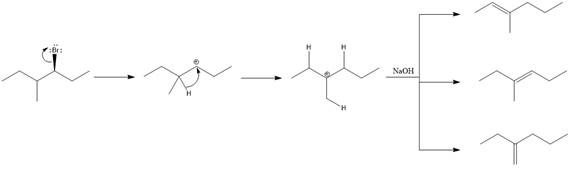
The major E1 product for the given reaction is shown below:
Explanation of Solution
Elimination usually takes place so as to produce the most stable, i.e., highly alkyl substituted, alkene.
The possible E1 products are obtained by eliminating the leaving group and a proton on carbon adjacent to the one bonded to the leaving group. Here the
From the stability of the product formed, the major E1 product is drawn.
(c)
Interpretation:
For the given substrate, when
Concept introduction:
As the number of alkyl groups on the carbon bonded to the leaving group increases, the rate of E1 reaction increases. Each additional alkyl group, which is electron donating, stabilizes the carbocation intermediate produced and thus helps the leaving group leave. The
Answer to Problem 9.69P
The major E1 product for the given reaction is shown below:
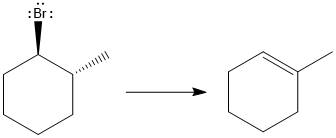
Explanation of Solution
Elimination usually takes place so as to produce the most stable, i.e., highly alkyl substituted, alkene.
The possible E1 products are obtained by eliminating the leaving group and a proton on carbon adjacent to the one bonded to the leaving group. Here the
From the stability of the product formed, the major E1 product is drawn.
(d)
Interpretation:
For the given substrate, when
Concept introduction:
As the number of alkyl groups on the carbon bonded to the leaving group increases, the rate of E1 reaction increases. Each additional alkyl group, which is electron donating, stabilizes the carbocation intermediate produced and thus helps the leaving group leave. The
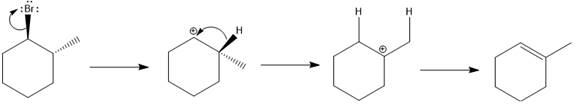
Answer to Problem 9.69P
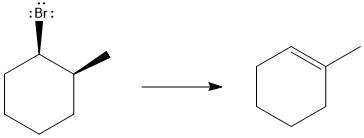
The major E1 product for the given reaction is shown below:
Explanation of Solution
Elimination usually takes place so as to produce the most stable that means highly alkyl substituted alkene.
The possible E1 products are obtained by eliminating the leaving group and a proton on the carbon adjacent to the one bonded to the leaving group. Here the
The first alkene product is highly substituted than the second alkene product. Therefore, the first alkene product is more stable. The base in this reaction is OH, which is not bulky, so the first and second products are the major products.

From the stability of the product formed, the major E1 product is drawn.
(e)
Interpretation:
For the given substrate, when
Concept introduction:
As the number of alkyl groups on the carbon bonded to the leaving group increases, the rate of E1 reaction increases. Each additional alkyl group, which is electron donating, stabilizes the carbocation intermediate produced and thus helps the leaving group leave. The
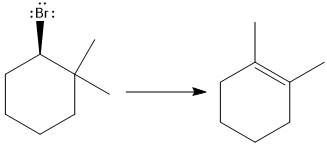
Answer to Problem 9.69P
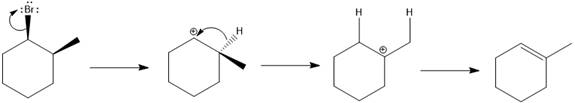
The major E1 product for the given reaction is shown below:
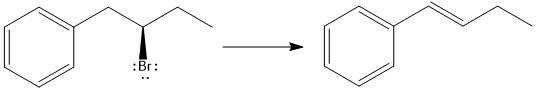
Explanation of Solution
Elimination usually takes place so as to produce the most stable that means highly alkyl substituted alkene.
The possible E1 products are obtained by eliminating the leaving group and a proton on the carbon adjacent to the one bonded to the leaving group. Here the
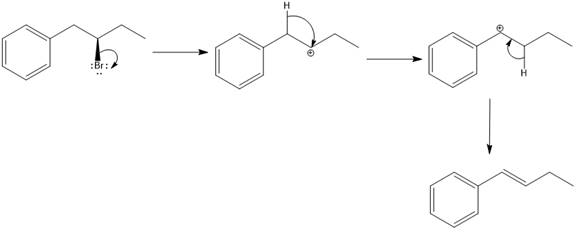
From the stability of the product formed, the major E1 product is drawn.
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry: Principles And Mechanisms: Study Guide/solutions Manual (second)
- The major product of the given reaction has the molecular formula CH03. Draw its structure in the most stable tautomeric form. Select Draw Rings More Erase H 1. NaOCH,CH3 2. H3O* CH;CH2OHarrow_forwardThe molecule, 2-bromo-1,3-diethyl-1,4-dimethylcyclohexane undergoes an SN1/E1 reaction in the solvent ethanol. i) Draw the intermediate resulting from the first step of this reaction. ii) Draw the two most favorable intermediates resulting from carbonation iii) From the intermediate(s) in (ii) draw all the possible E1 products and circle the product(s) that is(are) most favorable. iv) Name the most favorable product(s). v) Draw an SN1 product from an intermediate in (ii).arrow_forwardPlease propose a mechanism for the reaction which takes place under conditions that favor an SN1 reaction.arrow_forward
- Heating an alcohol in the presence of sulfuric or phosphoric acid will cause a dehydration to occur: the removal of the elements of water from a molecule, forming an alkene. The reaction usually follows an E1 mechanism. The SN1 pathway is suppressed, both by heating the reaction mixture (more energy promotes the elimination pathway) and by using a strong acid whose conjugate base is a poor nucleophile. Draw curved arrow(s) to show how the alcohol reacts with phosphoric acid.arrow_forwardPLEASE HELParrow_forwardGive two possible [1,2]-rearrangement products where an alkyl group migrates in the following compound. Motivate also which of the two products will be formed preferentiallyarrow_forward
- See image belowarrow_forwardThe following molecules are subject to substitution (SN1 or SN2) reaction conditions. a) Identify if the leaving group (-Br) is attached to a 3°, 2°, 1°, or methyl Carbon. b) Rank the molecule with respect to their SN1 reactivity, with 1 being the fastest and 4 being the slowest. c) Rank the molecule with respect to their SN2 reactivity, with 1 being the fastest and 4 being the slowest. Br Br CH3B Br a. type of Carbon on C-Br b. SN1 reactivity (1 fastest, 4 slowest) c. SN2 reactivity (1 fastest, 4 slowest)arrow_forward4) One of the diastereomeric cyclohexyl bromide starting materials below will readily undergo an E2 reaction while the other one will not. Fill in the requested information in boxes and lines indicated including the structure of the alkene product that does undergo an E2 reaction (use scratch paper first please). draw the expected major alkene product draw lowest E chair conformations Me. Ме Me EtO: Na Me "Ме :Br: Ме. Ме Me Me" Ме EtO: Na :Br: Required arrangement of H and LG (Br) for E2? (no acronyms). What is the rate law for and E2? On the template at left, draw all o & o* orbitals involved in the allowed E2 process.arrow_forward
- Each of the following may participate in an elimination reaction, under the proper conditions. (a) Circle the alpha (a) carbon. (b) Circle the beta (B) carbon(s). (c) Draw the alkene product(s) that may form, with the new double bond between the a and ß positions. (d) If more than one alkene product is possible, circle the most stable (Zaitsev) product. A В J C K E F G 33-0arrow_forwardDraw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any inorganic byproducts. H CH3 H Br Ph CH2CH3 NaNha Marrow_forwardConsider the SN2 reaction of butyl bromide with OH- ion. CH3CH2CH2CH2B + OH- → CH3CH2CH2CH2OH + Br Assuming no other changes, what effect on the rate would result from simultaneously doubling the concentrations of both butyl bromide and the OH- ion? Draw the curved arrow mechanism for the SN1 reaction of methanol with tert-butyl iodide. Next, draw an Energy vs. Reaction Coordinate diagram for this reaction, labeling all transition states as "TS" and intermediates as appropriate for those drawn in the mechanism above, showing the relative energies on the diagram.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
